 |
 |

Methicillin-Resistant Staphylococcus aureus Infections in 2 Pediatric Outpatients
Henry M. Feder, Jr, MD
Arch Fam Med. 2000;9:560-562.
ABSTRACT
Methicillin-resistant Staphylococcus aureus (MRSA) infections are an emerging problem in children. The following are 2 case reports of unsuspected MRSA infections: the first is an infant with cervical adenitis and the second is a child with a deep infection of the toe. Both patients failed outpatient therapy with oral cephalosporins and required hospitalization for surgical drainage. Both patients had cultures positive for MRSA at surgery. Neither patient had any risk factors for acquiring MRSA. Thus, outpatients with presumed staphylococcal infections who fail oral therapy with cephalosporins may be infected with MRSA.
INTRODUCTION
Community-acquired methicillin-resistant Staphylococcus aureus (MRSA) infections are unusual in children.1-6 The following are case reports of 2 unrelated children who were treated as outpatients for S aureus infections. Outpatient antibiotic therapy failed for these children and they were hospitalized. Both patients had unsuspected MRSA infections.
REPORT OF CASES
CASE 1
A 19-month-old girl was seen by her physician for fever and irritability. Physical examination revealed a 2 x 2-cm, tender, indurated anterior cervical node. Her temperature was 38.9°C (102°F). The patient was treated for what was thought to be a staphylococcal infection with oral cephalexin (250 mg 4 times daily). After 3 days of outpatient therapy, the node was enlarging and she was hospitalized at Connecticut Children's Medical Center, Hartford. On admission, her temperature was 40°C (104°F) and a 3 x 4-cm left cervical node was present. The node was tender, erythematous, and firm. It was not fluctuant. Her peripheral leukocyte count was 24.5 x 109/L with 0.12 bands and 0.65 polymorphonuclear neutrophils. Her erythrocyte sedimentation rate (ESR) was 45 mm/h. She was treated with intravenous cephazolin (100 mg/kg per day). On the third hospital day she was still febrile and the node was unchanged. She was then taken to the operating room where about 3 mL of purulent material was drained from the node. Gram stain revealed many neutrophils and rare Gram-positive cocci. For the next 2 days she remained febrile and irritable. The node remained painful and erythematous and there was no further drainage. Throughout the course of her illness, she remained alert and had no nuchal signs. On day 5 of hospitalization, the cultures from the node were positive for MRSA (Table 1) and therapy was changed to intravenous vancomycin. Her fever and irritability abated during the next 2 days. During the following 3 days the swelling, pain, and erythema of the node resolved. Vancomycin therapy was stopped after 10 days. Careful questioning of her family revealed no exposure to someone at risk for MRSA, including no hospital or nursing home exposures, no day care exposures, no exposures to someone who used intravenous drugs, and no exposure to someone with a staphylococcal infection. In addition, the patient had never received antibiotics prior to this illness. At follow-up 6 months later, the patient was normal except for a 1-cm-diameter, nontender, movable, firm node.
|
|

|
|
Break Point Minimum Inhibitory Concentration Susceptibilities for MRSA Isolates From 2 Pediatric Patients*
|
|
|
CASE 2
A 10-year-old girl presented to her physician with a tender, erythematous, swollen fourth toe. The swelling had been present for 1 week. There was no history of trauma or prior problems with the toe. The patient was afebrile. Her physical examination results were normal except for the distal two thirds of her toe, which was swollen, warm, erythematous, and mildly tender. No break in the skin was found. A radiograph was normal and she was treated with oral cephadroxil (500 mg twice daily) for cellulitis. After 10 days of therapy, her toe was unchanged and a second radiograph showed a lytic lesion of the middle phalynx of the fourth toe consistent with osteomyelitis. Her complete blood cell count and ESR were normal. She was hospitalized for debridement. On admission, she was afebrile and the distal two thirds of her toe was swollen about 50% greater than normal size. Her toe was erythematous and slightly tender but not warm. Her peripheral leukocyte count was 4.3 x 109/L with a normal differential. Her ESR was 8 mm/h. At surgery, the phalynx contained a small amount of purulent material, which was positive at culture for a pure heavy growth of MRSA (Table 1). She was treated with 4 weeks of intravenous clindamycin and did well. Treatment with oral clindamycin was attempted, but she was unable to tolerate the taste of the liquid and could not swallow the capsules. She had no exposure to someone at risk for MRSA and had not received antibiotics for the year prior to this illness. At follow-up, 3 months after completing therapy, her toe was normal except for the surgical scar.
COMMENT
Penicillin was introduced for the treatment of S aureus infections in the early 1940s. By the end of that decade, much of the S aureus (acquired both in hospitals or in the community) produced 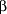 -lactamase and was resistant to penicillin.7 In the 1960s, semisynthetic penicillins (methicillin, oxacillin) and cephalosporins were developed, which were not inactivated by -lactamase and was resistant to penicillin.7 In the 1960s, semisynthetic penicillins (methicillin, oxacillin) and cephalosporins were developed, which were not inactivated by 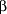 -lactamase.8 In 1961, MRSA was first described.9 The mechanism of resistance was a change in the penicillin (cephalosporin) binding proteins (PBPs) within the cell membrane. This change in the PBPs resulted in penicillins and cephalosporins binding less avidly to PBP 2a.10 Resistance of the PBPs to penicillins and cephalosporins was chromosomally mediated by the MEC-A gene.11 In the 1960s, MRSA were found sporadically throughout the United States and most MRSA isolates were sensitive to some orally administered antibiotics like clindamycin, macrolides, and tetracyclines.12 In the 1970s, MRSA resistant to orally administered antibiotics and sensitive only to vancomycin was reported.13 Since the 1970s, MRSA has become a nosocomial problem in hospitals for both children and adults. In adults, MRSA infection also occurs in nursing home patients, in intravenous drug users, and may be community acquired without risk factors.11-14 Methicillin-resistant S aureus may be sensitive to some antibiotics like clindamycin, macrolides, tetracyclines, trimethoprim/sulfamethoxazole, and quinolones, or it may be resistant to all antibiotics except vancomycin. Recently, MRSA resistance to all antibiotics, including vancomycin, has been reported.15 -lactamase.8 In 1961, MRSA was first described.9 The mechanism of resistance was a change in the penicillin (cephalosporin) binding proteins (PBPs) within the cell membrane. This change in the PBPs resulted in penicillins and cephalosporins binding less avidly to PBP 2a.10 Resistance of the PBPs to penicillins and cephalosporins was chromosomally mediated by the MEC-A gene.11 In the 1960s, MRSA were found sporadically throughout the United States and most MRSA isolates were sensitive to some orally administered antibiotics like clindamycin, macrolides, and tetracyclines.12 In the 1970s, MRSA resistant to orally administered antibiotics and sensitive only to vancomycin was reported.13 Since the 1970s, MRSA has become a nosocomial problem in hospitals for both children and adults. In adults, MRSA infection also occurs in nursing home patients, in intravenous drug users, and may be community acquired without risk factors.11-14 Methicillin-resistant S aureus may be sensitive to some antibiotics like clindamycin, macrolides, tetracyclines, trimethoprim/sulfamethoxazole, and quinolones, or it may be resistant to all antibiotics except vancomycin. Recently, MRSA resistance to all antibiotics, including vancomycin, has been reported.15
Reports of community-acquired MRSA infections have recently been reported in children.1-6 In 1995, Pate et al1 reported a case of MRSA septic arthritis in a 6-year-old boy from Mississippi. The route of acquisition of MRSA was unknown. Herold et al2 reviewed all cases of MRSA from pediatric inpatients at the University of Chicago Children's Hospital, Chicago, Ill. From 1988 through 1990, 8 (25%) of 32 cases of MRSA were community acquired and in 7 of these 8 patients an exposure to MRSA was elicited. From 1993 through 1995, 35 (67%) of 52 cases of MRSA were community acquired and 10 of these 35 patients had an identified exposure. Thus, in the second part of this study (1993-1995) of 52 pediatric patients hospitalized with MRSA, 25 (48%) patients had community-acquired MRSA without an identified exposure.2 Adcock et al3 described 2 infants from 2 separate day care centers in Texas who required hospitalization for pneumonia and empyema secondary to MRSA. Cultures of the other children at the 2 day care centers revealed that 2 (3%) of 61 children carried MRSA at one center and 9 (24%) of 40 children carried MRSA at the other center. In 1999, Shahin et al5 reported community-acquired MRSA otitis media in a 2-year-old child from Toronto, Ontario, and transmission to a day care contact with chronic dermatitis. Thus, MRSA can colonize at the day care setting. Lastly, MRSA has been identified as the cause of boils among 6 members of a high school wrestling team.14
During the 1998 calendar year, 14% of the S aurerus isolates recovered from both inpatients and outpatients at the Connecticut Children's Medical Center were MRSA; however, the epidemiology of these isolates was not studied. In a pilot project done by the Connecticut Department of Public Health, the epidemiology of S aureus blood isolates from New Haven was studied. Of approximately 100 patients with blood cultures positive for S aureus, 22 (22%) were MRSA. For at least 1 (4.5%) of those 22, no risk factors for acquiring MRSA were identified (personal communication, James L. Hadler, MD, MPH, State Epidemiologist).
Hospital-acquired MRSA is usually resistant to most oral antibiotics, including clindamycin, whereas community-acquired MRSA is usually sensitive to clindamycin. Some S aureus appears to be MRSA but the organisms really are high 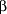 -lactamase producers and lack the MEC-A gene. These high -lactamase producers and lack the MEC-A gene. These high 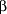 -lactamase producers are usually susceptible to clindamycin. Methicillin-resistant S aureus (in clindamycin-susceptible isolates) can be verified by the presence of the MEC-A gene, which was done in our second case.5 -lactamase producers are usually susceptible to clindamycin. Methicillin-resistant S aureus (in clindamycin-susceptible isolates) can be verified by the presence of the MEC-A gene, which was done in our second case.5
Most MRSA infections have been reported in adults with hospital exposures, nursing home exposures, or intravenous drug use and in children with hospital exposures. Community-acquired MRSA infections are a problem of adults and an emerging problem of children. We document 2 pediatric patients who failed outpatient antibiotic therapy because of unsuspected MRSA infections.
AUTHOR INFORMATION
Accepted for publication October 25, 1999.
Corresponding author: Henry M. Feder, Jr, MD, Connecticut Children's Medical Center, 282 Washington St, Hartford, CT 06106.
From Connecticut Children's Medical Center, Hartford.
REFERENCES
 |  |
1. Pate KR, Nolan RL, Bannerman TL, Feldman S. Methicillin-resistant Staphylococcus aureus in the community. Lancet. 1995;346:978.
2. Herold BC, Immergluck LC, Maranan MC, et al. Community-acquired methicillin-resistant Staphylococcus aureus in children with no identified predisposing risk. JAMA. 1998;279:593-598.
FREE FULL TEXT
3. Adcock PM, Pastor P, Medley F, Patterson JE, Murphy TV. Methicillin-resistant Staphylococcus aureus in two child care centers. J Infect Dis. 1998;178:577-580.
ISI
| PUBMED
4. Layton MC, Hierholzer HJ Jr, Patterson JE. The evolving epidemiology of methicillin-resistant Staphylococcus aureus at a university hospital. Infect Control Hosp Epidemiol. 1995;16:12-17.
ISI
| PUBMED
5. Shahin R, Johnson IL, Jamieson F, McGeer A, Tolkin J, Ford-Jones EL. Methicillin-resistant Staphylococcus aureus carriage in a child care center following a case of disease. Arch Pediatr Adolesc Med. 1999;153:864-868.
FREE FULL TEXT
6. Lindenbayer JM, Schoenfeld S, O'Grady R, Carney JK. Methicillin-resistant Staphyloccus aureus in a high school wrestling team and the surrounding community. Arch Intern Med. 1998;158:895-899.
FREE FULL TEXT
7. Berber M, Rozwadowska-Dowzenko M. Infection by penicillin-resistant staphylococci. Lancet. 1948;2:641-644.
PUBMED
8. Rolinson GN, Stevens S, Batchelor FR, Wood JC, Chain EB. Bacteriological studies on a new penicillin: BRL 1241. Lancet. 1960;2:564-567.
FULL TEXT
| PUBMED
9. Jevons MP. "Celbenin"-resistant staphylococci. BMJ. 1961;1:124-125.
10. Gutmann MM. Methicillin-resistant Staphylococcus aureus and vancomycin-resistant enterococci: therapeutic realities and possibilities. Lancet. 1997;349:1901-1906.
FULL TEXT
|
ISI
| PUBMED
11. Boyce JM. Are the epidemiology and microbiology of methicillin-resistant Staphylococcus aureus changing? JAMA. 1998;279:623-624.
FREE FULL TEXT
12. Brumfitt W, Hamilton-Miller J. Methicillin-resistant Staphylococcus aureus. N Engl J Med. 1989;320:1188-1196.
ISI
| PUBMED
13. Methicillin-resistant Staphylococcus aureus. Med J Aust. 1982;1:445-488.
PUBMED
14. Moreno F, Crisp C, Jorgensen JH, Patterson JE. Methicillin-resistant Staphylococcus aureus as a community organism. Clin Infect Dis. 1995;21:1308-1312.
ISI
| PUBMED
15. Smith TL, Pearson ML, Wilcox KR, et al. Emerence of vancomycin resistant Staphylococcus aureus. N Engl J Med. 1999;340:493-501.
FREE FULL TEXT
RELATED ARTICLE
The Archives of Family Medicine Continuing Medical Education Program
Arch Fam Med. 2000;9(6):551-552.
FULL TEXT
THIS ARTICLE HAS BEEN CITED BY OTHER ARTICLES
The effect of antibiotics on methicillin-resistant Staphylococcus aureus
Dancer
J Antimicrob Chemother 2008;61:246-253.
ABSTRACT
| FULL TEXT
|