 |
 |

Is Caffeine a Flavoring Agent in Cola Soft Drinks?
Roland R. Griffiths, PhD;
Ellen M. Vernotica, PhD
Arch Fam Med. 2000;9:727-734.
ABSTRACT
 |  |
Background Concern has been expressed about the nutrition and health impact of high rates of soft drink consumption. Caffeine is an added ingredient in approximately 70% of soft drinks consumed in the United States. The soft drink manufacturers' justification to regulatory agencies and the public for adding caffeine to soft drinks is that caffeine is a flavoring agent.
Objective To examine the claim that caffeine plays an integral role in the flavor profile of soft drinks, by examining the effect of caffeine on the threshold for detection of flavor differences in cola beverages.
Design Double-blind crossover study starting November 1998 and ending July 1999.
Setting An academic research center.
Participants Twenty-five adult regular consumers of cola soft drinks. Based on a screening session, all were able to detect a flavor difference between cola containing sugar and diet cola.
Intervention A sensitive version of a forced-choice flavor-detection procedure was used to evaluate the effects of a wide range of caffeine concentrations (range, 0.05-1.6 mg/mL) on the ability to detect flavor differences between caffeinated and caffeine-free cola beverages. Repeated tests permitted determination of significant detection at each concentration in individual subjects.
Main Outcome Measures Percentage of subjects significantly detecting a flavor difference and mean percentage of trials correct at each caffeine concentration.
Results Detection of flavor differences increased as a function of caffeine concentration. At the 0.1-mg/mL concentration, which is the approximate concentration in the majority of cola soft drink products, 2 subjects (8%) significantly detected a flavor difference and the mean percentage correct (53%) was at chance levels.
Conclusions The finding that only 8% of a group of regular cola soft drink consumers could detect the effect of the caffeine concentration found in most cola soft drinks is at variance with the claim made by soft drink manufacturers that caffeine is added to soft drinks because it plays an integral role in the flavor profile. It is valuable for the general public, the medical community, and regulatory agencies to recognize that the high rates of consumption of caffeinated soft drinks more likely reflect the mood-altering and physical dependence–producing effects of caffeine as a central nervous system–active drug than its subtle effects as a flavoring agent.
INTRODUCTION
IN 1942, the American Medical Association Council on Foods and Nutrition stated:
From the health point of view it is desirable especially to have restriction of such use of sugar as is represented by consumption of sweetened carbonated beverages and forms of candy which are of low nutritional value. The Council believes it would be in the interest of the public health for all practical means to be taken to limit consumption of sugar in any form in which it fails to be combined with significant proportions of other food of high nutritive quality.1
In 1998, Americans consumed 15 billion gallons (216 billion liters) of flavored carbonated beverages (ie, soft drinks), which is the equivalent of 585 twelve-oz (355-mL) cans for every man, woman, and child.2 Consumption of soft drinks in the United States has increased 9-fold since the American Medical Association expressed its concern about the nutritional impact of soft drinks, and consumption has more than doubled since 1975.3-4 Currently, soft drinks represent the single largest source of added sugar in the American diet5 and there is continuing concern that consumption of soft drinks, particularly by children and teenagers, adversely affects health by increasing sugar and calorie consumption and by displacing consumption of more healthful foods,3, 5 thereby possibly increasing the likelihood of tooth decay,6-7 obesity,3, 8 and bone fracture.3, 9-11 In some cases, adults and children become physiologically and psychiatrically dependent on caffeinated soft drinks, experiencing a withdrawal syndrome if they terminate regular use, and feeling compelled to continue consuming soft drinks despite desires and recommendations that they quit.12-16
Caffeine is an added ingredient in approximately 70% of soft drinks sold in the United States (calculated from consumption data2). In response to a proposal in 1981 by the US Food and Drug Administration (Rockville, Md) to delete caffeine from cola-type beverages,17 soft drink manufacturers justified adding caffeine to soft drinks on the basis that caffeine is a flavor enhancer.18 A similar justification for adding caffeine to citrus-flavored beverages has formed the basis of a recent petition19 by soft drink manufacturers to the Health Protection Branch of Health Canada, which is the Canadian food regulatory agency (Ottawa, Ontario). The petition asserted that caffeine is an integral part of the overall flavor profile of soft drinks, which have attained their popularity because they activate a variety of flavor sensations.20 A comparable rationale for the role of caffeine in soft drinks is currently provided to the general public by soft drink manufacturers and the National Soft Drink Association (Washington, DC).21-22 The argument that the high consumer appeal of caffeinated soft drinks is due to the important role of caffeine in the overall flavor profile of these beverages overlooks the central nervous system–mediated stimulating, reinforcing, and physical dependence–producing actions of caffeine that provide a compelling alternative explanation for the high rates of consumption of caffeinated soft drinks.23-25
Based on the published literature, it is difficult to assess the credibility of the claim that caffeine plays an integral role in the flavor profile of soft drinks. Although caffeine has been recognized as a bitter substance that can be used as a flavoring ingredient,26-27 the effect of caffeine on the detection of flavor differences in soft drinks has not been reported. Since 1981, the 2 most popular brands of cola soft drinks (Coca-Cola Classic® and Pepsi Cola®), which account for more than a third of all soft drinks consumed in the United States,2 have had caffeine concentrations of about 0.1 mg/mL or lower.28-31 Although average detection thresholds of caffeine in water have often exceeded this concentration,32-36 studies have also shown that detection thresholds for caffeine vary widely between individuals,32, 36-39 can be lowered with explicit taste detection training,38 and are raised in the presence of other tastes or flavors.35, 37 Furthermore, caffeine and similar bitter substances show complex interactions with other tastes. Research examining simple 2-substance or 3-substance systems has shown that the addition of a second or a third taste can either suppress or potentiate the other tastes, depending on the substances under study.35, 40-43 Thus, precise prediction of how a substance such as caffeine would affect the flavor profile of a chemically complex mixture such as a cola soft drink that probably contains hundreds of different flavors44 is beyond the current state of oral sensory science.
The purpose of this study was to examine the claim that caffeine plays an integral role in the flavor profile of soft drinks by examining the effect of caffeine on the threshold for detection of flavor differences in cola beverages. Because there are large individual differences in taste sensitivity to caffeine, and to maximize the generality of the findings to consumers of these beverages, the study was conducted with volunteers who regularly consume caffeinated colas and who reported a preference for cola brand and for caffeine content based on flavor. Also, to increase the generality of the findings to consumers at large, volunteers did not receive extensive training in flavor discrimination. Instead, volunteers were screened as being able to discriminate regular vs diet cola, and a sensitive procedure was then used to determine the flavor-detection threshold within individual subjects.
SUBJECTS AND METHODS
SUBJECTS
Participants were recruited to participate in a soda taste study through notices posted on public bulletin boards throughout a large academic medical center campus (Johns Hopkins Bayview, Baltimore, Md). After preliminary telephone screening about their cola consumption, prospective subjects reported to the research unit, where they completed a medical history questionnaire, their vital signs were taken, and they were interviewed about their cola consumption. Subjects were eligible for participation if they were aged 21 to 55 years; were medically healthy; and reported consuming a cola beverage at least once a week, having a preference for a specific brand of cola (eg, Coca-Cola Classic®) based on flavor and a preference for the caffeine content of soda (ie, a preference for caffeinated over noncaffeinated, or vice versa) based on flavor. The study was approved by the local institutional review board.
STUDY PROCEDURES
Of the 55 individuals who completed the preliminary telephone screening, 10 were not qualified and 4 were unwilling or unable to report to the research unit for further screening. Of the 41 individuals who reported for further screening, 4 were not qualified and 5 were unwilling or unable to participate in the first flavor-detection test session. Unknown to participants, the first session consisted of participants attempting to detect a flavor difference between a sugar-containing cola and a diet cola. The data from the first session were used to eliminate individuals who were relatively insensitive to flavor differences. Of the 32 individuals who participated in the first session, 7 failed to detect a flavor difference and were dropped from the study at this point. All of the remaining 25 eligible participants completed the subsequent 6 flavor-detection test sessions, which consisted of participants attempting to detect a flavor difference between caffeinated cola and caffeine-free cola over 6 caffeine concentrations.
FLAVOR-DETECTION PROCEDURE
The procedure used for detecting flavor differences (2-alternative, forced-choice test with warm-up; trial-by-trial feedback; and financial payment for correct responses) is sensitive and permits testing differences under conditions in which the dimension of the flavor difference cannot be specified in advance.45-47 Sessions, which were scheduled at the same time each day, consisted of 25 trials during each of which participants tasted and swallowed 2 samples (5- to 6-mL) of 2 different cola solutions (totalling approximately 275 mL throughout the session). The interval between trials and between samples within a trial was 30 seconds. Participants rinsed and swallowed bottled spring water (15-30 mL on each occasion; 750-1500 mL throughout the session) between trials and between samples within a trial. Bottled spring water (Naya; Naya Inc, Mirabel, Quebec) was chosen for the study because it was ranked the most neutral tasting of several bottled waters tested.48 Cola samples and water rinse were presented at room temperature (22°-24°C).
Within a session, the 2 samples were arbitrarily designated "sample A" and "sample B." The first 5 trials of the session were warm-up trials in which the participant was informed of the contents of each sample cup (eg, "This cup contains sample B") immediately before tasting. Each warm-up trial consisted of the presentation of both samples, with the participant tasting the sample on the left first. Order of samples was mixed across warm-up sessions. The final 20 trials of the session were test trials. As before, each trial consisted of the presentation of 2 samples, 1 of which was sample A and the other sample B. The participant tasted and swallowed the contents of both cups (left to right) and then guessed which cup contained sample A and which cup contained sample B. Immediately after each guess, the participant was told whether he or she was correct or incorrect. In addition to their session pay of $10 per session, participants were paid $0.25 for each correct guess. At the end of each session, participants provided a written description of the basis on which they thought sample A and sample B were different.
Participants and the research assistants who interacted with them were told that the purpose of the flavor-detection test procedure was to determine whether participants could discriminate between soft drinks containing different commonly used taste or flavor enhancers, including theophylline, quinine, sugar, caffeine, tannin, saccharin, aspartame, and phosphoric acid. For all participants, the first session consisted of attempting to detect a flavor difference between a sugar-containing cola vs a diet cola. Participants who failed to correctly detect a flavor difference in greater than or equal to 85% of 20 trials were dropped from the study at this point. The remaining participants completed 6 additional flavor-detection test sessions. Each session consisted of attempting to detect a flavor difference between a caffeine-containing cola vs a caffeine-free cola at a single caffeine concentration. Six caffeine concentrations, 0.05 mg/mL, 0.1 mg/mL, 0.2 mg/mL, 0.4 mg/mL, 0.8 mg/mL, and 1.6 mg/mL (ie, 50 to 1600 ppm or 0.0002523 to 0.0082934 mol/L), were tested during the 6 sessions. The sequence of concentrations was mixed across subjects. Sessions were generally conducted 3 to 5 times per week.
SAMPLE PREPARATION
For the first session (regular vs diet cola), samples were prepared from a commercially available cola containing high-fructose corn syrup and/or sucrose (Caffeine Free Coca-Cola Classic®) and a commercially available cola containing aspartame (Caffeine Free Diet Coke®). For each of the subsequent 6 sessions, caffeinated and caffeine-free cola samples were prepared from a single bottle of commercially available caffeine-free cola (Caffeine Free Coca-Cola Classic®). Samples were prepared from a single bottle to eliminate confounding by possible differences in flavor and/or carbonation between different bottles of cola. Caffeinated cola samples were prepared by adding anhydrous caffeine to the caffeine-free cola. Visual inspection indicated that the anhydrous caffeine dissolved completely within several hours of preparation. Caffeine-free samples were handled in the same way as caffeinated samples. All samples were prepared at least 1 day before sessions, stored in glass screw cap containers to maintain carbonation, and maintained at room temperature. Aliquots of the test samples (5-6 mL) were poured into 30-mL medicine cups immediately before sessions.
DATA ANALYSIS
Within-Subject Analysis
For each subject in each session, the ability to detect a flavor difference between the 2 cola conditions (regular vs diet cola in session 1; caffeine vs no caffeine in sessions 2-7) was analyzed using the binomial probability distribution. Flavor-detection performance was considered significant if the subject was correct on 15 or more of the 20 test trials (P<.05).
Group Analysis
For analysis of the mean percentage of trials correct for regular vs diet cola and for caffeine vs no caffeine at each of the 6 caffeine concentrations, 1-tailed independent t tests were conducted comparing the observed mean with a hypothesized population mean of 10 (ie, 50% or chance performance).
RESULTS
Twenty-five subjects completed the protocol (14 female, 11 male; 13 white, 2 Hispanic, 3 Asian, 7 black). Subjects had a mean age of 29.7 years (SD, 8.3 years). All had at least a high school diploma or equivalent, and 16 were college graduates. All were currently employed. Six were current cigarette smokers. Sixteen reported consuming some alcohol weekly (mean, 3.1 drinks per week for users). Subjects reported consuming cola beverages a mean of 5.6 days per week (SD, 1.5 cola beverages). Number of cola beverages consumed on a typical day was the equivalent of 2.3 (mean) 12-oz (355-mL) servings (range, 1-6 servings; SD, 1.3 servings). Estimated annual cola consumption, adjusted for any seasonal variation reported by subjects, was the equivalent of 752 (mean) 12-oz (355-mL) servings (range, 176-2184 servings; SD, 538 servings). In terms of brand flavor preference, 14 preferred Coca-Cola Classic®; 5, Pepsi®; 4, Diet Pepsi®; and 2, Diet Coke®. All but 1 subject reported preferring the flavor of caffeinated cola to caffeine-free cola.
Figure 1 shows the percentage of subjects significantly detecting flavor differences (top) and the mean percentage of trials correct (bottom) for each of the experimental conditions in the 25 subjects. The figure shows that all subjects correctly detected a flavor difference between diet and regular cola, with a mean percentage correct of 98%. The high detection accuracy in this condition reflects the fact that participants who were unable to discriminate diet vs regular cola at greater than or equal to 85% were dropped from the study. The figure also shows that both the percentage of subjects significantly detecting a flavor difference and the mean percentage trials correct increased as a function of caffeine concentration across the 6 concentrations. No subject was able to significantly detect a difference (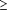 75%) at the lowest caffeine concentration (0.05 mg/mL) and the mean percentage correct was at chance levels (52%). At the 0.1 mg/mL concentration, 2 subjects (8%) significantly detected a flavor difference and the mean percentage correct remained at chance levels (53%). Increasing concentrations of caffeine (0.2 mg/mL, 0.4 mg/mL, 0.8 mg/mL, and 1.6 mg/mL) were significantly detected by 56%, 96%, 100%, and 100% of subjects, respectively, with the mean percentages correct being 74%, 95%, 99%, and 100%, respectively. 75%) at the lowest caffeine concentration (0.05 mg/mL) and the mean percentage correct was at chance levels (52%). At the 0.1 mg/mL concentration, 2 subjects (8%) significantly detected a flavor difference and the mean percentage correct remained at chance levels (53%). Increasing concentrations of caffeine (0.2 mg/mL, 0.4 mg/mL, 0.8 mg/mL, and 1.6 mg/mL) were significantly detected by 56%, 96%, 100%, and 100% of subjects, respectively, with the mean percentages correct being 74%, 95%, 99%, and 100%, respectively.
|
|

|
|
Effects of caffeine concentration on the ability to detect a flavor difference in a cola soft drink. The first bar shows the results of the screening session in which all subjects significantly detected a flavor difference between a sugar-containing cola vs a diet cola. The other bars show the results of sessions testing the detection of flavor differences between caffeinated cola and caffeine-free cola across 6 caffeine concentrations. Top, The percentage of subjects significantly detecting a flavor difference. Flavor detection was significant in a given subject if the subject was correct on 15 or more of the 20 test trials (P<.05). Bottom, Mean percentage trials correct. Bars show means; brackets show +1 SD; asterisks indicate which conditions were significantly different from chance (P<.05). The approximate caffeine concentration in the 2 most widely consumed cola soft drink products is 0.1 mg/mL.
|
|
|
At the end of each session, participants provided a written description of the basis on which they thought the samples were different. Those subjects who correctly detected a flavor difference between caffeine vs no caffeine at the lower concentrations (0.1 mg/mL and 0.2 mg/mL) most often described the caffeine samples as being "bitter" and/or as having an "aftertaste." Higher caffeine concentrations were often further described as being extremely unpleasant (eg, "like medicine" or "nasty").
COMMENT
This study shows that in a group of regular consumers of cola soft drinks, the effect of caffeine on the threshold for detection of flavor differences in a cola beverage is an orderly increasing function of caffeine concentration. At the caffeine concentration of 0.1 mg/mL, only 2 subjects (8%) could detect a flavor difference, and the overall data for the group was nonsignificant. This caffeine concentration is similar to or somewhat higher than that in the 2 most popular brands of cola soft drinks (Coca-Cola Classic® and Pepsi®),28-31 which account for more than half of cola soft drinks and more than one third of all soft drinks consumed in the United States.2 At 0.2 mg/mL, which is the highest caffeine concentration permitted in soft drinks by the Food and Drug Administration,49 56% of the subjects were able to detect a flavor difference. At this concentration, subjects usually described the caffeine-containing sample as bitter or as having an aftertaste. There is only one commercially marketed cola to have this level of caffeine (Jolt Cola; Wet Planet Beverages, Rochester, NY), whose marketing and advertising strategy is based not on superior flavor but on having "twice the caffeine." Overall, the results of the study are at variance with the claim made by soft drink manufacturers that caffeine is added to soft drinks because it plays an integral role in the flavor profile.
The subject population used in our study was chosen as most appropriate for examining the effect of caffeine on the flavor of cola soft drinks among usual consumers of those beverages. All subjects were regular cola drinkers who claimed that they already had a preference for either caffeinated cola to caffeine-free cola, or vice versa. All subjects also demonstrated flavor sensitivity by significantly discriminating regular cola from diet cola. In addition to the practice obtained in the screening session, the flavor-detection procedure involved explicit flavor-detection training during the warm-up and test trials in which trial-by-trial feedback with a financial contingency was provided. It is possible that more extensive flavor-detection training would have increased the sensitivity of subjects; however, such a demonstration would not be relevant to the question of whether a naturally occurring preference for cola drinks depends on the role of caffeine. Finally, although the study was conducted in adults, it is likely that similar results would have been obtained in children, many of whom are also regular consumers of soft drinks,3 since previous research has shown that caffeine detection thresholds are not lower in children than adults.36
The failure to detect a flavor difference at the caffeine concentration found in popular cola beverages (0.1 mg/mL) cannot be attributed to using an insensitive flavor-detection procedure. The study used a 2-alternative forced choice test with a warm-up procedure that is among the most sensitive of flavor-detection methods developed, and is appropriate for testing differences under conditions in which the dimensions of flavor difference cannot be specified in advance.45-47 The addition of trial-by-trial feedback with a financial payment for correct responses could be expected to further increase the sensitivity of the procedure.50 Other testing parameters (eg, sample volume, temperature, intersample interval, rinsing between samples, and sample swallowing requirement) are similar to those that have been used in previous research demonstrating low caffeine detection thresholds in aqueous solutions in certain individuals.32-33,36
Although the sample size in our study was only 25, it is sufficient to provide meaningful information about the detectability of caffeine in cola beverages. To our knowledge, almost all of the caffeine taste detection studies published to date used sample sizes smaller than 25 per group.32-35,37-39 In our study, the number of subjects detecting a difference and the mean percentage of trials correct were monotonically increasing functions of caffeine concentration, which demonstrates that the procedure used was sensitive to the manipulation of caffeine concentration. At the concentration of 0.1 mg/mL, which is similar to that delievered in most cola soft drinks, only 2 of 25 subjects detected a flavor difference, and the mean percentage of trials correct was only 53% (SD, 14.7%), which is very close to 50%, or chance levels. From these data, it seems that increasing the sample size would not have affected the conclusion that caffeine does not play an important role in the flavor of cola beverages for the large majority of regular cola consumers.
It is interesting that the subjects in our study claimed to have a preference for cola based partly on their belief that caffeinated cola tastes different from noncaffeinated cola. Furthermore, all but 1 of the subjects preferred caffeinated cola to caffeine-free cola, which would seem to be consistent with manufacturers' claims about the role of caffeine. It would be informative to know whether subjects could, in fact, detect differences in flavor under double-blind conditions. We did not attempt to assess the claim that caffeine contributes to the flavor profile of cola by directly comparing commercially available caffeine-containing cola with caffeine-free cola because the contents of those products are trade secrets and may differ on dimensions other than just caffeine. Related to this, a possible critique of our study is that we tested the role of caffeine by adding caffeine to a commercially available caffeine-free beverage (Caffeine Free Coca-Cola Classic®). It is possible that other changes in the formula of this beverage render it less sensitive to the effects of caffeine. Because the results of the present study have important implications for the justification of adding caffeine to noncola soft drinks (eg, Mountain Dew [PepsiCo Inc, Purchase, NY], Sunkist [Sunkist Growers Inc, Sherman Oaks, Calif]), it would also be informative to replicate our design using a broader range of flavored soft drink vehicles.
Caffeine's effects on the central nervous system provide a compelling alternative explanation for the strong preference for caffeine-containing over caffeine-free cola drinks claimed by participants in this study, and for the high rates of consumption of these caffeinated soft drinks. Caffeine is a stimulant drug that produces mild positive mood changes (eg, increases in feelings of energy and alertness)51-53 and is self-administered54-56 at doses delivered in soft drinks. With daily consumption of caffeine at a dose equivalent to that of 3 servings of cola soft drink (100 mg), caffeine has been shown to produce physical dependence characterized by lethargy and headache on cessation of intake.57-58 Suppression of low-grade withdrawal symptoms after overnight abstinence has been suggested as a principal mechanism by which caffeine engenders daily self-administration,24, 59-61 including preference for flavors paired with caffeine.24-25 In some cases, people become psychiatrically dependent on caffeinated soft drinks, feeling compelled to continue consuming soft drinks despite desires and recommendations that they quit.12, 14, 16
In addition to caffeine producing physical dependence and playing a prominent role in maintaining beverage consumption, various other caffeine-related health concerns have been recently expressed,62 including the displacement of nutritionally healthful foods by caffeinated sugar-containing soft drinks,3, 5 and possible adverse effects of caffeine in anxiety and sleep disorders63 and in pregnancy.64 In response to potential health concerns and the aggressive marketing of caffeinated soft drinks, in 1997 the American Medical Association adopted a resolution to work with the Food and Drug Administration to ensure that, when caffeine is added to a product, the amount of caffeine is displayed prominently on the label.65 The Food and Drug Administration is currently considering a petition from the Center for Science in the Public Interest (Washington, DC), which also calls for labeling the caffeine dose.66
In conclusion, the findings of our study suggest that despite claims to the contrary, caffeine plays a relatively minor role as a flavoring agent in cola soft drinks for the majority of regular consumers. We cannot, of course, rule out the possibility that some alteration in the testing protocol or parameters might permit development of a procedure uniquely sensitive to demonstrating, among normal users, a prominent role of caffeine as a flavoring agent in soft drinks. In the meantime, we believe that it is worthwhile for the general public, the medical community, and regulatory agencies to recognize the possibility that the high consumption rates of caffeine-containing soft drinks are more likely to reflect the mood-altering and physical dependence–producing effects of caffeine as a central nervous system–active drug than its subtle effects as a flavoring agent.
AUTHOR INFORMATION
Accepted for publication May 17, 2000.
This study was supported in part by grant R01 DA03890 from the US Public Health Service (Rockville, Md).
A preliminary version of this article was presented to The College on Problems of Drug Dependence, Acapulco, Mexico, June 15, 1999.
We thank Joseph Harrison, for technical assistance in conducting the study, Linda Felch, for consultation on statistical analysis, and Eric Strain, MD, Una McCann, MD, and George Bigelow, PhD, for their helpful comments on the manuscript.
Corresponding author: Roland Griffiths, PhD, Department of Psychiatry, Behavioral Pharmacology Research Unit, Johns Hopkins University School of Medicine, 5510 Nathan Shock Dr, Baltimore, MD 21224 (e-mail: rgriff{at}jhmi.edu).
From the Departments of Psychiatry and Behavioral Sciences (Drs Griffiths and Vernotica) Neuroscience (Dr Griffiths), Johns Hopkins University School of Medicine, Baltimore, Md.
REFERENCES
 |  |
1. American Medical Association, Council on Foods and Nutrition. Some nutritional aspects of sugar, candy, and sweetened carbonated beverages. JAMA. 1942;120:763-765.
FREE FULL TEXT
2. Beverage Digest Company. Beverage Digest Fact Book 1999. Bedford Hills, NY: Beverage Digest Co; 1999.
3. Jacobson MF. Liquid Candy: How Soft Drinks Are Harming America's Health. Washington, DC: Center for Science in the Public Interest; 1998.
4. Gilbert RM. Caffeine consumption. In: Spiller GA, ed. Methylxanthine Beverages and Foods: Chemistry, Consumption, and Health Effects. New York, NY: Liss; 1984:185-213.
5. Bowman SA. Diets of individuals based on energy intakes from added sugars. Fam Econ Nutr Rev. 1999;12:31-38.
6. Ismail AI, Burt BA, Ekland SA. The cariogenicity of soft drinks in the United States. J Am Dent Assoc. 1984;109:241-245.
ABSTRACT
7. Glynn K. The role of food and beverages in causing and preventing dental decay. The Canadian Soft Drink Association Web site. Available at: http://www.softdrink.ca/prw94en.htm#10.0. Accessed December 3, 1999.
8. Troiano RP, Briefel RR, Carroll MD, Bialostosky K. Energy and fat intake of children and adolescents in the United States: data from the National Health and Nutrition Examination Surveys. Am J Clin Nutr. In press.
9. Wyshak G, Frisch RE. Carbonated beverages, dietary calcium, the dietary calcium/phosphorous ratio, and bone fractures in girls and boys. J Adolesc Health. 1994;15:210-215.
FULL TEXT
|
ISI
| PUBMED
10. Wyshak G, Frisch RE, Albright TE, Albright NL, Schiff I, Witschi J. Nonalcoholic carbonated beverage consumption and bone fractures among women former college athletes. J Orthop Res. 1989;7:91-99.
FULL TEXT
|
ISI
| PUBMED
11. Wyshak G. Teen-aged girls, carbonated beverage consumption, and bone fractures. Arch Pediatr Adolesc Med. 2000;154:610-613.
FREE FULL TEXT
12. Strain EC, Mumford GK, Silverman K, Griffiths RR. Caffeine dependence syndrome: evidence from case histories and experimental evaluations. JAMA. 1994;272:1043-1048.
FREE FULL TEXT
13. Goldstein A, Wallace ME. Caffeine dependence in school children? Exp Clin Psychopharmacol. 1997;5:388-392.
FULL TEXT
|
ISI
| PUBMED
14. Hughes JR, Oliveto AH, Liguori A, Carpenter J, Howard T. Endorsement of DSM-IV dependence criteria among caffeine users. Drug Alcohol Depend. 1998;52:99-107.
FULL TEXT
|
ISI
| PUBMED
15. Bernstein GA, Carroll ME, Dean NW, Crosby RD, Perwien AR, Benowitz NL. Caffeine withdrawal in normal school-age children. J Am Acad Child Adolesc Psychiatry. 1998;37:858-865.
FULL TEXT
|
ISI
| PUBMED
16. Bernstein GA, Carroll ME, Thuras P, Cosgrove K, Monroe M, Winters K. Caffeine dependence in adolescents. In: Scientific Proceedings 45th Annual Meeting of the American Academy of Child and Adolescent Psychiatry. Washington, DC: American Academy of Child and Adolescent Psychiatry; 1998:106.
17. Caffeine: deletion of GRAS status, proposed declaration that no prior sanction exists, and use on an interim basis pending additional study. Federal Register. 69817-69838 (1980).
18. PepsiCo, Inc. The physical or technical effect of caffeine in cola beverages, July 20, 1981. In: Volume III, Appendix XII of Comments of the National Soft Drink Association submitted to the Department of Health and Human Services Food and Drug Administration in Response to the Proposal to Delete Caffeine in Cola-Type Beverages From the List of Substances Generally Recognized as Safe and to Issue an Interim Food Additive Regulation Governing Its Future Use. July 29, 1981. FDA Docket No. 80N-0418.
19. Government of Canada, Department of Health. Regulations Amending the Food and Drug Regulations (1079). Canada Gazette. Part I. January 3, 1998.
20. Pepsi-Cola Canada Beverages. Use of Caffeine as a Flavoring Agent in Non-Cola Carbonated Soft Drinks, Volume 1: Food Additive Submission for the Health Protection Branch. Ottawa, Ontario: Health Canada; 1996. Health Canada submission ADDFS96100901.
21. The Coca-Cola Company. Top Ten Answers to the Ten Most Frequently Asked Questions About Our Soft Drinks. Atlanta, Ga: Coca-Cola Co; 1997.
22. National Soft Drink Association. What's in Soft Drinks. Available at: http://www.nsda.org/softdrinks/History/whatsin.html. Accessed November 17, 1999.
23. Griffiths RR, Mumford GK. Caffeine–a drug of abuse? In: Bloom FE, Kupfer DJ, eds. Psychopharmacology: The Fourth Generation of Progress. New York, NY: Raven Press Ltd; 1995:1699-1713.
24. Rogers PJ, Richardson NJ, Elliman NA. Overnight caffeine abstinence and negative reinforcement of preference for caffeine-containing drinks. Psychopharmacology. 1995;120:457-462.
FULL TEXT
| PUBMED
25. Yeomans MR, Spetch H, Rogers PJ. Conditioned flavour preference negatively reinforced by caffeine in human volunteers. Psychopharmacology. 1998;137:401-409.
FULL TEXT
| PUBMED
26. Hall RL, Oser BL. Recent progress in the consideration of flavoring ingredients under the food additives amendment, III. GRAS substances. Food Technol. 1965;253:151-197.
27. Brieskorn CH. Physiological and therapeutical aspects of bitter compounds. In: Rouseff RL, ed. Bitterness in Foods and Beverages. New York, NY: Elsevier; 1990:15-33.
28. Consumers Union. Caffeine: how to consume less. Consumer Report. 1981;46:597-599.
29. Stavric B, Klassen R. Caffeine content in colas from New York State and Ontario. J Food Safety. 1987;8:179-185.
30. Galasko GTF, Furman KI, Alberts E. The caffeine contents of non-alcoholic beverages. Food Chem Toxicol. 1989;27:49-51.
FULL TEXT
|
ISI
| PUBMED
31. Mumford GK, Benowitz NL, Evans SM, et al. Absorption rate of methylxanthines following capsules, cola and chocolate. Eur J Clin Pharmacol. 1996;51:319-325.
FULL TEXT
|
ISI
| PUBMED
32. Mela DJ, Mattes RD, Tanimura S, García-Medina MR. Relationship between ingestion and gustatory perception of caffeine. Pharmacol Biochem Behav. 1992;43:513-521.
FULL TEXT
|
ISI
| PUBMED
33. Mattes RD. Influences on acceptance of bitter foods and beverages. Physiol Behav. 1994;56:1229-1236.
FULL TEXT
| PUBMED
34. Schiffman SS, Gatlin LA, Frey AE, Heiman SA, Stagner WC, Cooper DC. Taste perception of bitter compounds in young and elderly persons: relation to lipophilicity of bitter compounds. Neurobiol Aging. 1994;15:743-750.
FULL TEXT
|
ISI
| PUBMED
35. Schiffman SS, Gatlin LA, Sattely-Miller EA, et al. The effect of sweeteners on bitter taste in young and elderly subjects. Brain Res Bull. 1994;35:189-204.
FULL TEXT
|
ISI
| PUBMED
36. James CE, Laing DG, Oram N. A comparison of the ability of 8-9-year-old children and adults to detect taste stimuli. Physiol Behav. 1997;62:193-197.
FULL TEXT
| PUBMED
37. Mackey AO, Valassi K. The discernment of primary tastes in the presence of different food textures. Food Technol. 1956;10:238-240.
38. Pangborn RM. Influence of hunger and sweetness preferences on taste thresholds. Am J Clin Nutr. 1959;7:280-287.
FREE FULL TEXT
39. Hall MJ, Bartoshuk LM, Cain WS, Stevens JC. PTC taste blindness and the taste of caffeine. Nature. 1975;253:442-443.
FULL TEXT
| PUBMED
40. Pangborn RM. Taste interrelationships. Food Res. 1960;25:245-256.
41. Schiffman SS, Diaz C, Beeker TG. Caffeine intensifies taste of certain sweeteners: role of adenosine receptor. Pharmacol Biochem Behav. 1986;24:429-432.
FULL TEXT
|
ISI
| PUBMED
42. Calviño AM, García-Medina MR, Cometto-Muñiz JE, Rodríguez MB. Perception of sweetness and bitterness in different vehicles. Percept Psychophys. 1993;54:751-758.
ISI
| PUBMED
43. Breslin PAS, Beauchamp GK. Salt enhances flavour by suppressing bitterness. Nature. 1997;387:563.
PUBMED
44. Pendergrast M. The sacred formula [appendix]. In: For God, Country and Coca-Cola. New York, NY: Charles Scribner's Sons; 1993:421-425.
45. Ennis DM. Relative power of difference testing methods in sensory evaluation. Food Technol. April 1990:114-118.
46. O'Mahony M, Thieme U, Goldstein LR. The warm-up effect as a means of increasing the discriminability of sensory difference tests. J Food Sci. 1988;53:1848-1850.
FULL TEXT
|
ISI
47. Thieme U, O'Mahony M. Modifications to sensory difference test protocols: the warmed up paired comparison, the single standard duo-trio and the A-not-A test modified for response bias. J Sens Stud. 1990;5:159-176.
48. Kemp SE, Beauchamp GK. Flavor modification by sodium chloride and monosodium glutamate. J Food Sci. 1994;59:682-686.
FULL TEXT
|
ISI
49. Food and Drug Administration. Caffeine. Washington, DC: US Government Printing Office, Code of Federal Regulations Title 21, Food and Drugs, Part 182.11180; 1998.
50. Breslin PAS, Beauchamp GK, Pugh EN Jr. Monogeusia for fructose, glucose, sucrose, and maltose. Percept Psychophys. 1996;58:327-341.
ISI
| PUBMED
51. Lieberman HR, Wurtman RJ, Emde GG, Coviella ILG. The effects of caffeine and aspirin on mood and performance. J Clin Psychopharmacol. 1987;7:315-320.
ISI
| PUBMED
52. Griffiths RR, Evans SM, Heishman SJ, et al. Low-dose caffeine discrimination in humans. J Pharmacol Exp Ther. 1990;252:970-978.
FREE FULL TEXT
53. Silverman K, Griffiths RR. Low-dose caffeine discrimination and self-reported mood effects in normal volunteers. J Exp Anal Behav. 1992;57:91-107.
FULL TEXT
|
ISI
| PUBMED
54. Silverman K, Mumford GK, Griffiths RR. Enhancing caffeine reinforcement by behavioral requirements following drug ingestion. Psychopharmacology (Berl). 1994;114:424-432.
FULL TEXT
| PUBMED
55. Hughes JR, Oliveto AH, Bickel WK, Higgins ST, Badger GJ. The ability of low doses of caffeine to serve as reinforcers in humans: a replication. Exp Clin Psychopharmacol. 1995;3:358-363.
FULL TEXT
56. Liguori A, Hughes JR, Oliveto AH. Caffeine self-administration in humans: 1. Efficacy of cola vehicle. Exp Clin Psychopharmacol. 1997;5:286-294.
FULL TEXT
|
ISI
| PUBMED
57. Griffiths RR, Evans SM, Heishman SJ, et al. Low-dose caffeine physical dependence in humans. J Pharmacol Exp Ther. 1990;255:1123-1132.
FREE FULL TEXT
58. Evans SM, Griffiths RR. Caffeine withdrawal: a parametric analysis of caffeine dosing conditions. J Pharmacol Exp Ther. 1999;289:285-294.
FREE FULL TEXT
59. Hughes JR, Oliveto AH, Bickel WK, Higgins ST, Badger GJ. Caffeine self-administration and withdrawal: incidence, individual differences and interrelationships. Drug Alcohol Depend. 1993;32:239-246.
FULL TEXT
|
ISI
| PUBMED
60. Schuh KJ, Griffiths RR. Caffeine reinforcement: the role of withdrawal. Psychopharmacology. 1997;130:320-326.
FULL TEXT
| PUBMED
61. Garrett BE, Griffiths RR. Physical dependence increases the relative reinforcing effects of caffeine versus placebo. Psychopharmacology. 1998;139:195-202.
FULL TEXT
| PUBMED
62. James JE. Understanding Caffeine: A Biobehavioral Analysis. Thousand Oaks, Calif: Sage Publications; 1997.
63. Strain EC, Griffiths RR. Caffeine. In: Tasman A, Kay J, Lieberman JA, eds. Psychiatry. Vol 1. Philadelphia, Pa: WB Saunders Co; 1997:779-794.
64. Eskenazi B. Caffeine – filtering the facts. N Engl J Med. 1999;341:1688-1689.
FREE FULL TEXT
65. American Medical Association. Caffeine Drinks (Resolution 523). Adopted by the American Medical Association House of Delegates at the June 1997 annual meeting.
66. Center for Science in the Public Interest. Petition for Amendment of Food-Labeling Regulations to Require Quantitative Labeling of Caffeine Content and Request for Review of Health Effects of Caffeine: Petition to the Food and Drug Administration. Washington, DC: Center for Science in the Public Interest; July 31, 1997. FDA Docket No. 97P-0329/CP1.
RELATED ARTICLES
Editor's Comment
Marjorie A. Bowman
Arch Fam Med. 2000;9(8):734.
FULL TEXT
The Archives of Family Medicine Continuing Medical Education Program
Arch Fam Med. 2000;9(8):735.
FULL TEXT
THIS ARTICLE HAS BEEN CITED BY OTHER ARTICLES
Caffeine Consumption and Weekly Sleep Patterns in US Seventh-, Eighth-, and Ninth-Graders
Pollak and Bright
Pediatrics 2003;111:42-46.
ABSTRACT
| FULL TEXT
|