The GoLoco Motif: Heralding a New Tango Between G Protein Signaling and Cell Division
- Department of Pharmacology Lineberger Comprehensive Cancer Center Unc Neuroscience Center The University of North Carolina at Chapel Hill Chapel Hill NC 27599-7365 USA
- DPS. E-mail dsiderov{at}med.unc.edu; fax 919-966-5640.
Abstract
The Gα and Gβγ components of heterotrimeric G proteins, typically associated with cell-surface receptor signaling, also partake in the macromolecular interactions that underlie cell polarity and cell division. Proteins with Gα-binding GoLoco motifs, such as Drosophila melanogaster Pins (for Partner of Inscuteable) and its mammalian counterpart LGN, participate in multi-protein complexes that maintain cellular asymmetry and orderly segregation of chromosomal content and daughter cell bodies. The GoLoco motif was recently identified as a selective Gα-binding partner: the GoLoco–Gα interaction can displace Gβγ and inhibit guanine nucleotide release from the bound Gα subunit. Recent x-ray crystallographic studies suggest ways in which GoLoco-motif peptides may modulate heterotrimeric G protein signaling. Such peptides could be exploited to help dissect the signals that underpin cell polarity and cell division processes.
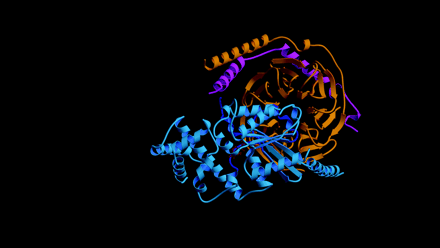
Heterotrimeric G proteins regulate signaling pathways downstream of plasma membrane-bound receptors. Several mechanisms have
been discovered that promote the separate effector functions of the Gα and the Gβγ subunits. Recent efforts have identified GoLoco motif-containing proteins as Gα-associated proteins that can regulate the reassociation of Gα to Gβγ, and that might effect signal transduction on their own. In what physiological processes are these proteins involved?
- © American Society for Pharmacology and Experimental Theraputics 2002



