Recombinant Pharmaceuticals from Plants: The Plant Endomembrane System as Bioreactor
Abstract
The production of safe pharmaceuticals at affordable costs is one of the great challenges of our times. Research has proven that transgenic plants can fulfill this need. This review focuses on the peculiar features of plant cells that allow high accumulation of recombinant proteins. The endomembrane system and the secretory pathway of plant cells in themselves offer a fascinating model of protein sorting, and in practical terms, represent the potential for the facile and very low-cost purification of recombinant pharmaceutical proteins.
Introduction
Plants have been used for thousands of years as sources of food and therapeutic agents. Well-known examples are the seeds of cereals and legumes, which for millennia have constituted the basis of nutrition, and the secondary metabolites of many plants that have been widely used in medicine. In recent decades, genetic engineering has dramatically opened the possibility of extending the variety of molecules of nutritional, therapeutic, and industrial importance that might be produced by plants. For a number of reasons, including production costs, safety, and environmental issues, plants may in the future become a major bioreactor of choice for producing recombinant proteins. The properties of plants as biofactories versus those of microorganisms, animal cell cultures, or transgenic animals have been evaluated in recent reviews (1–3). Here, we discuss in detail some specific features of plant cells that can make them, by virtue of their intriguing secretory pathway, particularly suitable for the production of high amounts of foreign proteins.
Thousands of animal and plant genes encode secretory proteins (4–5), which in animals include molecules of great medical importance, such as all milk proteins, all forms of immunoglobulins and the other blood serum proteins, digestive enzymes, peptide hormones and neurotransmitters, and collagen. With the exception of a few proteins synthesized in mitochondria and plant plastids, protein synthesis initiates in the cytosol. Secretory proteins have an N-terminal transient signal peptide and/or transmembrane domains; when the first of these sequences emerges from the ribosome, the whole translation complex is targeted to the endoplasmic reticulum (ER) translocation channel, where cotranslational insertion into the ER lumen or integration into the ER membrane occurs (6). These proteins are then either delivered to one of the compartments of the endomembrane system, which includes the ER, the Golgi complex, and hydrolytic compartments, or are secreted from the cell. Delivery occurs via vesicular traffic or by direct connections between the compartments; the major biosynthetic route, termed the secretory pathway (7, 8), is from the ER, through the Golgi complex, and then either to the hydrolytic compartments (i.e., vacuoles in plants and yeast or lysosomes in animal cells) or to secretion from the cell. Secretory proteins often undergo post-translational modifications that in many cases are necessary for their activity and stability. Examples are the formation of disulfide bonds between cysteine residues (9), N-glycosylation (10, 11), and specific proteolytic maturation steps (11, 12). Although exceptions exist (13, 14), recombinant secretory proteins will have to navigate the endomembrane system of eukaryotic host cells.
In all eukaryotes, soluble proteins that enter the ER and lack additional signals traffic to the Golgi complex and are then secreted from the cell by default (7, 8, 15). Otherwise, sorting to specific compartments of the endomembrane system requires specific signals or chemicophysical features. Therefore, important recombinant secretory proteins, such as antibodies, can be secreted (by default) from plant cells into the cell wall and the intercellular space (collectively termed the apoplast), where indeed high accumulation can be obtained (16, 17). A further advantage of plant biotechnology is that roots can release secreted proteins directly from the plant: the recombinant products can therefore be recovered from the water of hydroponically cultivated plants. This process of root secretion, termed “rhizosecretion” (18), obviates many purification procedures and allows constant recovery of product through the whole life cycle without the need to harvest the plant. Secretion, however, is merely the final phase of a complex process that involves a number of specific endomembrane features, each of which may in itself offer powerful opportunities for the accumulation of recombinant proteins. These opportunities, along with the endomembrane features that support them, are indicated in Figure 1⇓.
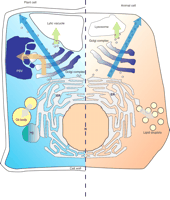
The secretory pathways of plant cells versus animal cells: Opportunities to exploit plants as bioreactors. Secretory proteins enter the ER (delimited here by the broken line, indicating membrane-bound ribosomes) in both cell types and travel to the Golgi complex via vesicular trafficking. In the absence of ER-retention signals, soluble proteins are then secreted (blue arrows). Vesicular sorting to the inner hydrolytic compartments (lytic vacuoles in plants, lysosomes in animals; green arrows) requires signals that differ in plants and animals. In addition to lytic vacuoles, plant cells may also have protein storage vacuoles (PSV), which develop according to specific signals and protein sorting mechanisms (orange arrow) and may be useful in the biotechnological production of recombinant pharmaceutical proteins. Vast amounts of proteins of cereals (e.g., the prolamin storage proteins) form protein bodies (PB) within the ER in aggregation processes that may also be exploited for biotechnology; the aggregation mechanisms are an area of active research and may involve prolonged interactions with chaperones, the formation of inter-chain disulfide bonds, and selective insolubility (28). Oil bodies and lipid droplets originate from the ER by the deposition of lipids in the space between the two monolayers of the ER membrane (gray arrows).
The Plant ER: A Protein Storage Compartment
In both plants and animals, the ER has two main functions: it allows entry into the secretory pathway and ensures that newly synthesized proteins fold and assemble correctly (15, 19). As mentioned above, secretory proteins enter the ER cotranslationally, in an unfolded state. The neutral pH, the oxidizing redox conditions, and the presence of numerous folding helpers optimize folding and assembly within the ER so that incorrectly folded proteins do not proceed along the secretory pathway.
For most secretory proteins, the ER is a compartment of transit. A number of severe genetic human diseases result from the abnormal accumulation within the ER of proteins that fail to traffic along the secretory pathway. These ER storage disorders are mainly due to mutations in cargo proteins (20). As a result of mutation, the proteins fail both to traffic and to be disposed by the ER protein quality control mechanism (19), resulting in large aggregates within this compartment that in the long term are toxic to cellular functions (20).
Conversely, the ER of plant cells can tolerate unusually high accumulation of protein without compromising plant development and reproduction, and the mechanisms through which naturally occurring resident proteins are retained within plant ER have been exploited (21). In all eukaryotes, proteins that remain resident in the ER have a C-terminal tetrapeptide, usually KDEL or HDEL, which is recognized by a receptor located in the Golgi complex (22). Upon binding, the receptor retrieves C-terminal tagged proteins back into the ER. The mechanism is therefore a retrieval rather than a true retention system, but it is very efficient. When the tetrapeptides are transferred by genetic engineering to other proteins, provided that a signal peptide for entry into the ER is added (if not already present), they promote in most cases ER retention. Localization within the ER via the addition of KDEL or HDEL increases by one or two orders of magnitude the accumulation of foreign protein in transgenic plants, a very desirable biotechnological achievement (21), indicating that the compartment has very low hydrolytic activity and is very plastic. This strategy has been used for the high accumulation of immunoglobulins, vaccines, and diagnostics in transgenic plants (23–26).
Plants naturally use the ER to accumulate vast amounts of protein in specific tissues, in the form of large aggregates or oligomers, as part of the developmental process of seed maturation (27, 28). The cargo proteins are some of the major storage proteins of the seeds of cereals such as maize, wheat, and rice, and have obvious great nutritional importance. They belong to the prolamin class of storage proteins, which are not found in organisms other than plants (29, 30). Prolamins do not have the KDEL or HDEL ER-localization signals. They form large aggregates within the ER lumen, termed protein bodies (Figure 1⇑). The molecular interactions that lead to the ER retention of prolamins and to protein body formation are still not fully clear, but a number of prolamins form protein bodies also when transiently synthesized in animal cells (31, 32) or permanently expressed in vegetative (non-seed) tissues of transgenic plants (33, 34), indicating that no peculiar machinery is needed for their accumulation within the ER.
Portions of γ-zein, a prolamin of maize, induce protein body formation and high accumulation of foreign protein within the ER when fused to proteins otherwise destined to other locations within the cell (Figure 2⇓; 35, 36). The γ-zein sequences involved can therefore be used as an alternative to the KDEL signal to accumulate recombinant proteins in the ER of plant cells (Figure 3⇓). A comparison between the effects of KDEL and the N-terminal half of γ-zein on the accumulation of the model protein phaseolin (i.e., a protein of common bean that is naturally stored in the vacuole rather than the ER) is informative (35). In transgenic leaf tissue, the phaseolin-zein fusion accumulates to 3.5% of total leaf protein, whereas the phaseolin–KDEL fusion reaches only 0.5%; wild-type phaseolin accumulates to less than 0.1% in transgenic vegetative tissues. There is thus a marked increase in protein stability by addition of the ER-retention signals, with the zein sequence significantly improving stability even when compared to the already quite stable KDEL-fusion. But given that both recombinant proteins accumulate within the ER, what is the mechanism that further increases the stability of the zein fusion over the KDEL-fusion? A tentative explanation is that the formation of protein bodies, mediated by the zein fragment but not by KDEL, excludes the protein from the normal, albeit relatively slow, processes of physiological turnover of ER resident proteins. Indeed, the stability of the protein bodies that plants produce within the ER is a remarkable function of plant developmental physiology. In cereals, for example, when the seed tissue that has served a nutritive function (i.e., the endosperm) to the developing “embryo” undergoes programmed cell death at the end of seed maturation, the ER within the “dying” tissue can nevertheless store a vast amount of protein that is excluded from the otherwise apoptotic physiology of this compartment. Intriguingly, this remarkable process can be reproduced for biotechnological purposes in vegetative plant tissues without compromising ER functions. This utility exploits the plasticity of plant cells, a property that may reflect their totipotency. Plants can be regenerated from single vegetative cells, and plant tissue differentiation can in many cases be reversed (37).
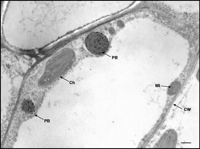
A domain of a maize storage protein can be used recombinantly to form ER-located protein bodies. Electron micrograph of a thin section of a young leaf of transgenic tobacco that expresses “zeolin,” a fusion between the N-terminal domain of the maize prolamin γ-zein and the vacuolar protein phaseolin. The recombinant protein forms electron-dense protein bodies (PB) not normally found in leaf cells. Zeolin was visualized with antibodies against phaseolin and secondary antibody-gold complex (black dots inside the PBs). The large empty area that occupies most of the cell is the lytic vacuole. [Ch, chloroplast; Mt, mitochondrion; CW, cell wall. Bar = 500 nm. Reproduced from (35).]
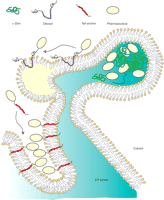
Strategies to accumulate high amounts of recombinant pharmaceuticals in the ER and ER-derived oil bodies. Through the use of plant protein domains (e.g., zein domains, oleosins, or tail-anchors), multiple protein fusions have been examined. Zein domains allow protein body formation within the ER lumen. Oleosin fusion products are first inserted into the external monolayer of the ER bilayer and then integrated into a nascent oil body (yellow). The tail anchor “anchors” recombinant proteins to the ER surface; upon overexpression, the ER can be commandeered in this way to develop distinct topologies that increase the stability of recombinant proteins. The hydrophobic domains of oleosin and of tail-anchored proteins are indicated in violet and red, respectively. Note that the N-terminal and C-terminal domains of oleosin are in the cytosol, and the very short C-terminal sequence of the tail-anchor is instead luminal.
Purification of recombinant proteins is an important issue in biotechnology, both in terms of costs and final productivity (2). The use of γ-zein fragments to accumulate recombinant proteins provides an advantage in this respect, because the protein bodies are insoluble (35) and therefore easily purified by centrifugation. The introduction of a consensus sequence for in vitro cleavage by proteases between the zein portion and the protein of interest can allow for further purification of the latter (36).
Oil Bodies for the High Accumulation and Easy Purification of Recombinant Proteins
Research on the mechanism used by oil seeds to store their lipid reserves has also provided an efficient strategy for the production and purification of recombinant proteins. Oil bodies are organelles that originate from the ER and function to store plant seed oil (Figure 1⇑). Like lipid droplets in animal and yeast cells, oil bodies are delimited by a phospholipid monolayer that is in direct contact with the lipid content (38). The oil body membrane contains a very abundant protein termed oleosin. Although they are targeted to the ER membrane using the same mechanism as other proteins, oleosins have a peculiar topology. A highly conserved hydrophobic central domain is embedded into the oil body matrix, whereas the N- and C-terminal domains are exposed to the cytosol on the surface of the oil body; this unique topology is the result of a “proline knot” in the hydrophobic domain, which contains three conserved proline residues (39).
Proteins expressed in the seeds of transgenic plants as translational fusions with oleosins are correctly targeted to and tightly associated with the oil body, remaining exposed to the cytosol (Figure 3⇑) (40). Oil bodies and proteins associated with them can be easily separated from the majority of other seed cell components by floatation centrifugation, taking advantage of their very low density (40). To facilitate the recovery of the pure product, a specific protease cleavage site can be inserted between oleosin and the pharmaceutical protein. This strategy has been used to produce a biologically active form of the thrombin inhibitor hirudin in transgenic seeds of oilseed rape (Brassica napus) (40). A further development of this technology is the use of engineered oil bodies as low-cost affinity matrices for the purification of target pharmaceuticals from biological extracts. The method involves the recombinant fusion between oleosin and a ligand, such as an immunoglobulin, having affinity for the desired target molecule (41).
Targeting Recombinant Proteins to the ER without Accessing the Lumenal Environment of the Secretory Pathway
A number of proteins with pharmaceutical properties are not secretory proteins, and their expression in the endomembrane system could result in undesired post-translational modifications, such as N-glycosylation, which could negatively affect protein function. Expression in the cytosol, however, potentially exposes the protein to degradation by the ubiquitin/proteasome system, and recombinant proteins in the cytosol usually accumulate to levels that are well below 0.1% of total protein, whereas in the secretory pathway they easily reach more than 1% (42–44).
The fusion with oleosin exposes the recombinant protein to the cytosol, therefore avoiding post-translational modifications that occur in the lumen of the endomembrane system; at the same time, association with the oil body seems to provide good protection from cytosolic degradation. An alternative strategy is to anchor the recombinant protein to the cytosolic face of the ER membrane via the C-terminal region of tail-anchored membrane proteins (TA proteins; also termed type IV membrane proteins) (45). TA proteins lack an N-terminal signal peptide and reach their destination by post-translational mechanisms. They are bound to the membrane by a C-terminally located hydrophobic sequence. This is followed by a short polar region that is the only part translocated in the lumen of the organelle of destination. In response to unusually elevated levels of TA proteins, the ER can be transformed into stacked membrane arrays (Figure 3⇑) that represent a new ER-derived compartment that could accommodate recombinant TA proteins (46). We have tested this innovative strategy by fusing the TA domain of the mammalian ER cytochrome b5 to potential recombinant antigens. The results indicate that these molecules are more stable than the cytosolic counterparts (E. Pedrazzini, unpublished results) and may provide a good strategy for preventing the rapid degradation of recombinant proteins, thus improving yield.
The Peculiar Features of Plant Vacuoles
All eukaryotic cells have inner acidic hydrolytic compartments, which are constituted by vacuoles in plants and yeast, and by lysosomes in animals (Figure 1⇑). The sorting of proteins to these compartments occurs at the Golgi complex and is mediated by specific protein receptors (47, 48). Plant vacuoles are also involved in the maintenance of cell turgor and in the storage of secondary metabolites and proteins (49). The accumulation of unmetabolized molecules in lysosomes, on the other hand, is a pathology, called lysosomal storage disorder, and results from genetic defects in lysosomal proteins (50).
Protein storage in vacuoles occurs mainly in seeds. Indeed, most seed storage proteins are vacuolar, and the accumulation of seed protein in the ER is more a peculiar feature of cereals than a rule (29). Two aspects of the biogenesis and functions of plant vacuoles are relevant for the production of recombinant proteins. First, the high hydrolytic activity of vacuoles of vegetative tissues must be considered, and second, the mechanisms of deposition of vacuolar storage proteins must be understood.
Protein Degradation in Vacuoles: Problems and Possible Solutions
To date, tobacco is the most widely used plant bioreactor for the production of pharmaceuticals in research laboratories (2). In the future, tobacco may also be a reactor of choice for large-scale production, owing to the ease of transformation and the high productivity. Tobacco biomass is constituted by leaves, which also provide the target tissue in certain plants, such as lettuce, that are under study for the direct administration of recombinant pharmaceuticals as food product without the need of purification (43). Leaf vacuoles are mainly of the lytic type and can degrade foreign protein, resulting in a loss of product. This problem has been observed in studies of a hybrid immunoglobulin A/G produced in transgenic tobacco. Although most of the recombinant immunoglobulin is secreted into the extracellular space (i.e., the apoplast), a portion is delivered to the vacuole and degraded (51). Deletion analysis of the hybrid molecule indicates that vacuolar delivery is mediated by the third constant domain of the IgA heavy chain, whereas the parent IgG similarly expressed in tobacco is quantitatively secreted (51, 52). Most likely, the pertinent IgA domain contains a motif that is recognized as a vacuolar sorting signal in plant cells (52); vacuolar sorting in plants is mediated by short amino-acid sequences that are very often contained in propeptides removed upon vacuolar delivery. There is not a canonical consensus and the signals are very diverse (48, 53). One class of vacuolar sorting receptors (the BP-80 class) has been identified. The mechanism is different with respect to animal lysosomes: lysosomal enzymes are sorted via the mannose-6-phosphate receptor, which recognizes phosphorylation of their glycan side chains (47); this system does not exist in plants. Therefore, the possible loss of foreign protein introduced into the plant secretory pathway due to vacuolar delivery must be verified experimentally for each individual protein, and it is very difficult to make predictions. In the case of a positive answer it may be possible to avoid vacuolar delivery by deleting or mutagenizing the signal (52). Alternatively, a method has been recently developed to inhibit protein sorting to plant vacuoles. This involves the transient expression of a codominant-negative chimera containing the transmembrane domain and the cytosolic tail of a member of the BP-80 receptor family (54). This leads to saturation of the recycling mechanism of the receptor with consequent depletion of BP-80 from the Golgi complex. As a result, proteins that normally interact with this receptor are secreted (54). This should also avoid vacuolar delivery of foreign proteins that fortuitously have sequences recognized by BP-80. Knock-outs of genes involved in the biogenesis of plant vacuoles are lethal or have altered growth (55–57) indicating that a permanent severe defect in vacuole biogenesis is not tolerated by plants, as expected based on the important functions of plant vacuoles. Therefore, for recombinant protein production, the expression of the BP-80 codominant-negative chimera should most probably be put under the regulation of easily inducible but tightly controlled promoters, to be activated only shortly before collection of material. Vacuolar delivery is however not always detrimental for protein production in plants, as we explain below.
Storage Vacuoles for Protein Accumulation
In the seeds of most plants, including the economically important grain legumes and oil seeds, storage proteins accumulate in vacuoles termed protein storage vacuoles (PSVs). The difference between PSVs and the “typical” vegetative vacuoles is not simply due to the presence of storage proteins in the former. PSVs originate de novo during seed maturation, and the mechanisms of protein sorting to the two types of vacuoles are, at least in part, distinct (Figure 1⇑) (53). Young cells that are not yet differentiated often have both types of vacuoles, and during differentiation either of the two types becomes prevalent, depending on the role that the cell ultimately assumes. PSVs have a slightly higher pH and lower hydrolytic activity than vacuoles of vegetative tissues (58).
PSVs of seeds are therefore an attractive compartment of accumulation for recombinant proteins or peptides. In a pioneering experiment, the DNA sequence coding for the pentapeptide Leuenkephalin was inserted, flanked by tryptic cleavage sites, into the “2S storage protein” of Arabidopsis thaliana (59). The peptide replaced five amino acids present in a region that is not conserved in storage proteins of the same class and is therefore probably not fundamental for correct folding. Transgenic oilseed rape plants were produced, and Leuenkephalin could be prepared from seeds by obtaining a protein fraction enriched in the recombinant 2S storage protein and subsequent protease digestion followed by chromatography (59).
Recent experiments suggest that it may not always be necessary to perform sophisticated protein engineering to allow targeting and accumulation of recombinant proteins in seed storage compartments. Human lysozyme, which is a secreted protein, accumulates in ER-derived protein bodies and in PSVs when expressed in the endosperm of rice seeds (Figure 4⇓) (60). Recombinant lysozyme produced in rice seeds is biologically active (61).
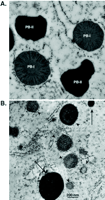
Human lysozyme is sorted into protein bodies and storage vacuoles in transgenic rice seeds. Electron micrographs of developing seed tissue (i.e., endosperm) of non-transgenic A. and transgenic B. rice seeds. In non-transgenic tissue A. the storage proteins accumulate in ER-derived protein bodies (PB-I) and in protein storage vacuoles (PB-II). In transgenic rice expressing human lysozyme B. the recombinant protein (visualized by immunogold labeling) accumulates in both PB-I-like (arrowheads) and PB-II-like (arrows) structures. Notice the ribosomes decorating the membrane of PB-I, indicating the ER origin. [Reproduced from (60).]
Rice naturally contains both ER-stored and vacuolar storage proteins; it is therefore possible, although not proved, that the high synthesis of these proteins has a dominant effect on the localization of foreign proteins expressed in the secretory pathway, perhaps because the aggregation properties of storage proteins (53) can influence traffic in the endomembrane system. The dominant effect of storage proteins on subcellular localization has also been observed in transgenic wheat. Both human serum albumin engineered to contain the C-terminal KDEL ER-localization signal and Asperigillus niger phytase (another secreted protein) are delivered to protein storage vacuoles together with storage proteins when expressed in wheat endosperm (62). In the leaf cells of the same transgenic plants (62), the human serum albumin–KDEL construct efficiently accumulates in the ER, and phytase expressed in tobacco leaves is secreted (63). Therefore, the two proteins have the subcellular localization expected from their molecular properties when expressed in vegetative plant tissues, confirming that vacuolar delivery in wheat endosperm was tissue-specific.
Conclusions
The expression of foreign proteins in the plant secretory pathway is perhaps the most promising technology for the high production of recombinant pharmaceutical at a cost that will be affordable worldwide. The peculiar features of the plant endomembrane system have been used in research laboratories to produce very high amounts of a few model proteins and pharmaceuticals. The advantages of using ER-derived protein bodies or oil bodies include the ready isolation of these structures and purification of associated proteins, along with the economic feasibility of simplified production. These technologies are being rapidly refined, and companies such as ERA Plantech in Barcelona and SemBioSys Genetics in Calgary have been specifically created to exploit them. The molecular mechanisms that give rise to oil bodies and ER protein bodies are still not fully understood, and research into tail-anchored proteins is at an even earlier stage. A more detailed characterization and the identification of key regulatory steps in the trafficking of proteins in plants will be invaluable for future developments. Specialized proteins such as oleosins and γ-zein and possibly others may prove very useful in genetic engineering, and the machineries involved in their accumulation (e.g., chaperones and other folding factors, as well as receptors involved in protein trafficking), have shown promise for biotechnological purposes. The use of PSVs is also promising, and indeed the first seed crop with improved nutritional quality has taken advantage of the introduction of a new vacuolar storage protein (64). Research on this, however, is at present less developed, perhaps also because of the reluctance to use seeds of major crops for the production of pharmaceuticals, given the possibility of contamination with the food chain. Possible strategies to overcome these problems include male sterile plants and cultivars and varieties, not normally used in agriculture, that produce seeds with easily distinguishable characteristics that will guard against contamination. Ongoing research into the molecular mechanisms of the biogenesis of PSVs (53) will also be of great help in designing strategies for the best exploitation of these compartments.
Acknowledgments
We thank Ning Huang for the pictures in Figure 4⇑. Work supported in part by the EU Integrated Project “Pharma-Planta” (LSHBCT-2003-503565).
- © American Society for Pharmacology and Experimental Theraputics 2005
References

Emanuela Pedrazzini, PhD, is a Junior Scientist at the Istituto di Biologia e Biotecnologia Agraria of the Consiglio Nazionale delle Ricerche in Milano, Italy. She is a plant cell biologist. Her main interest is in the molecular mechanisms of targeting and sorting of tail-anchored membrane proteins. Recently, she has developed a strategy to bind recombinant pharmaceuticals to intracellular membrances by fusing them with the tail-anchoring domains from TA proteins.

Alessandro Vitale, PhD, is a Senior Scientist at the Istituto di Biologia e Biotecnologia Agraria of the Consiglio Nazionale delle Ricerche in Milano, Italy. He has a long-term interest in the protein secretory pathway of plant cells and its involvment in the synthesis of seed storage proteins. More recently, his research has also focused on the exploitation of the secretory pathway to produce recombinant pharmaceuticals in plants. Address correspondence to AV. E-mail vitale{at}ibba.cnr.it; fax +39 02 23699 411.