HANGING IN THE BALANCE
KIR and Their Role in Disease
Abstract
The killer cell immunoglobulin-like receptors (KIR) are a recently discovered family of activating and inhibitory receptors that control natural killer (NK) cell function. KIR exist as a diverse family of receptors that have evolved rapidly by both gene duplication and recombination events. These findings were unexpected for a family of genes involved primarily in the innate immune response. These findings together with the observation that several of these genes have human leukocyte antigen (HLA) class I ligands, have led to a flurry of investigation into how KIR participate in viral infections, autoimmune diseases and malignancies. This review summarizes the major features of these genes and discusses how they may be involved in both disease pathogenesis and its amelioration.
Introduction
The immune system is a complex network of cellular and humoral components that has the primary function of discriminating self from non-self––such as, pathogens or altered self, including tumors––and then mounting the appropriate response to destroy them. One important component of this system are the Natural Killer (NK) cells. Originally NK cells were defined based on their morphology as large granular lymphocytes, with the ability to lyse target cells in a major histocompatibility complex (MHC) class I–independent manner, without the need for prior antigen exposure (1). In addition to this function, NK cells can secrete a wide variety of cytokines, including those typically secreted by T helper (Th) type 1 (Th1) and Th2 cells, and can interface directly with cells of the adaptive immune system including dendritic cells and T cells (2–6) (Box 1). Thus, the functions of NK cells are complex and include destruction of abnormal cells, and the shaping of the downstream adaptive immune response. Underpinning the key functions of NK cells is their ability to directly recognize abnormal cells and to generate a rapid response.
The Innate and Adaptive Immune System: A Comparison
Innate
-
▪ Evolutionarily ancient: Found in both animals and plants
-
▪ Immediate response to pathogens
-
▪ No memory for pathogens
-
▪ Non-rearranging genes for cell surface receptors
Adaptive
-
▪ Evolutionarily more recent: predominantly found in verterbrates
-
▪ Delayed primary response to pathogens
-
▪ More rapid and focussed response on rechallenge
-
▪ Receptors have genes that rearrange (T-cell and B-cell receptors)
Note: This reductionist view undermines the true complexity of the immune system. Frequently, immune cells and their receptors no longer sit neatly within these definitions.
The involvement of NK cells in disease processes is far-ranging. NK cells can lyse specific tumor targets that are deficient in a MHC class I cell surface marker (1), a molecule present on all human nucleated cells and known to be especially important in the immune response to viral infections (to discriminate self from non-self) (7). For example, the immune response to model pathogens such as murine cytomegalovirus (MCMV) is critically dependent on NK cells’ responses (8), and this finding has been extended to human viral infections, particularly of the herpes family (9, 10). Of note is that the Herpesviridae have evolved complex mechanisms for interfering with the cell biology of MHC class I recognition (11). NK cells, however, may also be involved in generating successful immune responses to other classes of viral infections whose immune evasion strategies may not be so intimately linked with MHC class I (12–14).
Based on key observations of NK function, the critical role of MHC class I in NK cell biology was formulated in the “missing-self” hypothesis (15). This concept of recognition proposed that NK cells lyse tumor targets that do not express self MHC class I. Thus, their ability to distinguish self from non- or altered self––an ability that T cells also possess––is, like CD8+ T cells, also based on MHC class I, but paradoxically it is the absence of this molecule that triggers the NK cell response. Thus, healthy cells express normal levels of MHC class I and NK cells recognize this molecule on the cell surface. This interaction generates an inhibitory signal, which prevents NK cells from lysing the target. In disease states that down-regulate surface expression of MHC class I––such as, tumors or viral infections––the lack of inhibitory signal prevents inhibition, and permits activation (16, 17). Furthermore, inhibition can also be overcome by the generation of a strong activation signal. Indeed, tumors can increase the expression of ligands for activating receptors on NK cells, and this may be sufficient to overcome the inhibitory signal that prevents NK cells from killing healthy autologous cells (18, 19). Thus, NK cell functions are determined by a complex network of activating and inhibitory receptor-ligand interactions, which are integrated at a cellular level to generate either a net inhibitory signal in health, or a net activating signal in disease. It is the regulation of this balance of signals that may be amenable to pharmacological intervention for the purpose of disease amelioration.
NK Cell Receptors: Generating Diversity
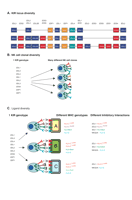
Unlike other lymphocytes, NK cells do not rearrange germ-line encoded receptors, which might suggest that NK cells are able to respond to only a limited a number of stimuli. However, NK cells possess a number of mechanisms that permit responses to a broad range of different infections and tumors: 1) they express many different receptors each of which may have a different ligand, which may be up- or down-regulated as a consequence of different discrete stimuli; 2) some receptors are expressed only on subsets of NK cells; and, 3) within the human population there is a genetic diversity of NK cell receptors, the most diverse of which are the killer-cell immunoglobulin-like receptors (KIR). A further level of diversity is conferred on the KIR by their HLA class I ligands, which also exhibit population diversity. This can lead to the relatively common situation in which, at a genetic level, an individual has a KIR but no ligand for that KIR (Figure 1⇓).
KIR generate diversity at multiple levels. A. The population diversity of KIR. A schematic is shown of the gene content illustrating three of the many possible different KIR haplotypes. Activating genes are in red, inhibitory genes in blue, and pseudogenes are in orange. B. Clonal expression of KIR. Shown are four potential NK cells derived from a single individual. KIR expression is clonal and in this illustration the individual NK cells express different combinations of KIR. C. Diversity in the HLA Class I ligands for KIR generate a functional KIR diversity. Illustrated are the functional effects of different HLA class I types in three individuals with the same KIR genotype. Individual A has four potential KIR:HLA inhibitory interactions, individual B has three, and individual C has two. All individuals have the NKG2A:HLA-E receptor ligand combination which exhibits very limited population diversity. Thus, the number of potential different inhibitory receptor:ligand interactions in each individual is different.
The expression of a wide range of receptors (Table 1⇓) with diverse ligands implies that NK cells can respond to many different stimuli. Many of these activating receptors are constitutively expressed on all NK cells and so it is the increased expression of their ligands on other cells by discrete stimuli that underpins activating receptor diversity. One much studied example is that of NKG2D (18). This well-conserved receptor is expressed on all NK cells and binds to a diverse range of ligands, some with as little as 20% amino acid identity (20). The expression of these ligands can be induced by stress, where different stressors induce different ligands (18, 21). Thus, one NK cell receptor allows the NK cell to respond to several different stimuli. The expression of other receptors, such as NKp44, can be increased by cytokine stimulation, implying that the NK cell receptor repertoire can be further modulated by exogenous factors (22).
Activating and Inhibitory Receptors on Human NK Cells
The isolation of the antibodies EB6 and GL183 by Moretta and colleagues (23, 24) that recognize cell surface molecules expressed predominantly on NK cells demonstrated that only subpopulations of human NK cells expressed these determinants. These molecules were subsequently identified as products of the KIR genes KIR2DL1 and KIR2DL3, respectively (23, 24). This observation was followed by studies in the mouse demonstrating a homologous receptor family to KIR, termed Ly49, whose individual gene products were also expressed only on subpopulations of NK cells (25), revealing that receptor expression on NK cells is variegated. NK cells, therefore, do not express all their germ line–encoded receptors, but rather, selected combinations of these are expressed in a stochastic manner. The pattern of Ly49 receptor expression is determined by a “probalistic switch” in the promoter regions of the Ly49 genes and a similar mechanism may operate for KIR (26). Thus, within a given individual there are subpopulations of NK cells that express different combinations of cell surface–activating and–inhibitory receptors, generating an NK cell repertoire in which different NK cell clones have the potential to respond to a wide variety of different stimuli (Figure 1⇑).
NK cells share many common features with T cells, including the expression of certain receptors. Most receptors expressed on NK cells are not unique to this cell lineage, nor, indeed, to the innate immune system. In fact, KIR are expressed on a subset of effector memory T cells, and members of the NKG2 family, especially NKG2D, are expressed on cytotoxic T cells and γδ-T cells (18, 27). Although this review will focus predominantly on the role of receptors on NK cells, some comments on their role on T lymphocytes will also be made.
KIR
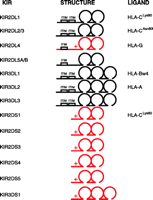
The KIR are members of the immunoglobulin superfamily of receptors and are encoded on chromosome 19q13.4 as part of the chromosomal region designated the leukocyte receptor cluster (LRC) (28). They are named according to their structural characteristics (Figure 2⇓). Thus, a KIR with two extracellular Ig domains and a short intracytoplasmic tail is named KIR2DS and given a number as a suffix, such as KIR2DS2 or KIR2DS4. A KIR with three extracellular Ig domains and a long intracytoplasmic tail is named KIR3DL, for example KIR3DL1 or KIR3DL2. These structural characteristics correlate with function. KIR with short intracytoplasmic tails are thought to activate NK cells via the adapter molecule DAP12 and those KIR with long intracytoplasmic tails inhibit NK cells via recruitment of SHP1 or SHP2 to their immunoreceptor tyrosine inhibitory motifs (ITIMs) (29–31).
Schematic of the different expressed KIR, their domain structures and their HLA class I ligands. The KIR in black are inhibitory and have immunoreceptor tyrosine inhibitory motifs (ITIMs). The KIR in red are activating and have a charged amino acid residue in the transmembrane region, illustrated with a “+” which permits them to pair with a signalling adaptor molecule. KIR2DL4 transduces an activating signal, but also has a single ITIM. The pseudogenes are not shown.
The specificity of the inhibitory KIR for their HLA class I ligands has been clearly defined in some cases (Figure 2⇑). KIR2DL1 binds the subset of HLA-C allotypes with lysine at position 80 of the heavy chain (HLA-CLys80); KIR2DL2 and KIR2DL3 bind the remaining HLA-C allotypes, which have asparagine at position 80 (HLA-CAsn80) (32, 33); KIR3DL1 recognizes HLA-B allotypes with a specific combination of amino acids at positions 77 to 83 of the heavy chain that combine to form the Bw4 serological motif (HLA-Bw4) (34, 35), which represents about one third of all HLA-B alleles, and KIR3DL2 binds HLA-A3 and -A11 (36, 37). The specificities of the activating KIR have been harder to define, as they seem to have a much lower affinity for putative HLA class I ligands. Based on sequence similarity, KIR2DS1 and KIR2DS2 should have the same HLA class I ligands as KIR2DL1 and KIR2DL2, respectively; however, KIR2DS1 binds HLA-C only weakly, and consistent binding of KIR2DS2 to HLA-C has not been shown (38). Furthermore, the surface expression on NK cells of KIR3DS1, an activating receptor with >97% sequence identity to KIR3DL1 in the extracellular domains (and, putatively, recognizing the same ligands), has not been demonstrated. Finally, KIR2DL4 has a long intracytoplasmic tail with ITIMs and interacts with the adaptor molecule FcεRIγ (39) and appears to transduce an activating signal (40). KIR2DL4 may also bind HLA-G in vitro (41), but the physiological role of this interaction remains unclear.
The complexity of KIR is further compounded by substantial allelic and haplotypic diversity. Each KIR gene has between four (KIR2DS1) and nineteen (KIR3DL1) alleles (42). Additionally, individuals can have anywhere from nine to sixteen KIR genes and these genes can occur together in many different combinations on a single chromosome to generate enormous haplotypic diversity (43, 44). This extreme degree of haplotypic diversity has been generated by a series of recombination and unequal crossing over events (45, 46). KIR diversity is also compounded at a functional level by diversity in the HLA ligands for KIR. The MHC locus in which the HLA class I ligands for KIR reside is on chromosome 6 and, thus, the KIR and its ligands segregate independently at meiosis. As KIR recognize only subsets of HLA class I molecules, a given individual may have a specific KIR, but may lack the corresponding HLA class I ligand; therefore, in a given individual, a specific KIR may be nonfunctional. For example, in the UK Caucasian population, approximately 34% have a nonfunctional KIR2DL1 gene owing to the absence of HLA-CLys80, whereas 15% have nonfunctional KIR2DL2 or KIR2DL3 genes owing to the absence of HLA-CAsn80(47).
Most human NK cells express not only an inhibitory receptor for self-MHC class I, but also express another transmembrane protein that comes from the NKG2 family of receptors (48). These receptors heterodimerize with CD94 to form a functional receptor, except for NKG2D, in which the functional molecule is a homodimer. Expression of the NKG2 genes, with the exception of NKG2D, is also clonal on NK cells (49); however, in contrast to the KIR, the NKG2 family of genes is well-conserved throughout evolution and exhibits little polymorphism or population diversity (50) and all individuals are thought to have all the NKG2 genes. These genes encode receptors of the C-type lectin-like family and exist in inhibitory forms (NKG2A) or in activating forms (NKG2C, NKG2D, and NKG2E). In humans, their ligands include the non-polymorphic HLA-class I molecule HLA-E (which binds to NKG2A, -C, or -E, heterodimerized with CD94) (51–53); MHC class I chain-related protein A (MIC-A), MIC-B; or the UL16 binding proteins (ULBPs) (all of which bind to a NKG2D homodimer) (18, 54). In the mouse, NKG2A and NKG2C recognize the functional homolog of human HLA-E, termed Qa-1 (55), and NKG2D binds products of the Retinoic Acid Early Inducible genes and H60 (21).
Overall, the activities of the KIR and NKG2 families of receptors synergize to generate the final NK cell repertoire, and expression of these receptors on individual NK cells appears relatively stable, as does the overall NK cell receptor repertoire of an individual (49, 56). However, NK cells that express high levels of NKG2A may have a particular role in directing the adaptive immune response, as it is this subpopulation that appears important for interacting with dendritic cells (DCs) and also for cytokine secretion (57, 58). Thus, the KIR and the NKG2 families are two gene families that generate diversity at population (KIR only) and clonal (KIR and NKG2) levels.
Genetic Studies of KIR and Disease
Genetic studies of the association of KIR with disease have been concerned mainly with viral infections and autoimmune disease. Indeed, the haplotypic diversity of KIR has made the KIR gene locus a compelling target for disease association studies. Comparison of humans with other primate species has revealed an unexpectedly rapid evolution of KIR consistent with pathogen-mediated selection (59). The expression of KIR on NK cells implies, therefore, that KIR are likely to be involved in determining the outcome of viral infections. Furthermore, the association of the MHC with autoimmune disease has suggested that specific KIR may also be involved in this group of diseases.
Viral infections
The key role of NK cells in viral infections has led to the investigation of KIR and their HLA class I ligands, especially in Human Immunodeficiency Virus (HIV), Hepatitis C Virus (HCV) and Cytomegalovirus (CMV) infections.
HIV
HIV is a viral infection causing immunosupression and progression to acquired immunodeficiency syndrome (AIDS) over a variable period of time. Analysis of HLA-B alleles in HIV-infected individuals has shown that the presence of homozygosity for HLA-Bw4––the ligand for KIR3DL1 and putatively KIR3DS1––is associated with a slower decline in CD4+ T-cell count, a marker for disease progression in HIV infection (60). Furthermore, analysis of over 1,000 individuals infected with HIV demonstrated that those with the compound genotype KIR3DS1 and a subset of HLA-Bw4+ alleles that have isoleucine at position 80 of the MHC class I heavy chain (Bw4Ile80) progress more slowly than those without this activating KIR-HLA combination (61). Indeed, KIR3DS1 in the absence of Bw4Ile80 was not protective, indicating the synergistic effect of these two alleles. To date, however, surface expression of KIR3DS1 has not been convincingly demonstrated; nevertheless, in functional experiments, it appears that Bw4Ile80 may be stronger than Bw4+ HLA-BThr80 as a ligand for KIR3DL1 allotypes, which would be consistent with the genetic observation. The effect of HLA-Bw4 as a factor delaying progression to AIDS was found in a small study of 88 individuals. In this study, a protective effect was observed in individual with an allele belonging to the HLA-B*57 supertype, a grouping that includes the HLA-B57 alleles together with the closely related HLA-B58 allele. Again, those alleles with isoleucine at position 80 were protective, but in this group there was a marginal increase in protection if this was found in combination with the inhibitory receptor KIR3DL1 (62). The numbers of individuals with KIR3DS1 were too small for meaningful analysis, but the difference found between this study and the study of Martin et al. (61) may reflect study design, study size, linkage disequilibrium, or the allelic diversity of KIR and HLA in different populations.
In HIV infection, it is not clear whether it is expression of KIR3DS1 on NK cells or on T cells that functionally correlates to the genetic observations. This conundrum may, in part, arise from a lack of suitable reagents to detect the expression of KIR3DS1; however, studies of expression of other KIR in HIV infection have shown changes in the T cell rather than the NK cell compartment (63, 64). The signal generated via the T-cell receptor can be modulated by KIR-HLA interactions, so that the expression of KIR by HIV-specific cytotoxic T-cells can alter cell sensitivity to HIV-derived peptides (65). The activating KIR3DS1-Bw4Ile80 interaction might result in increased activation of T cells, or NK cells, expressing KIR3DS1, resulting in direct lysis of infected target cells or recruitment of other cells of the immune system. Interestingly, HIV downregulates the expression of HLA-B (66); however, in vitro studies of the two–Ig domain KIR suggest that lower levels of cognate antibody, and by inference ligand, are required to trigger the activating receptors than their inhibitory counterparts (67). Thus, the genetic findings in this study are compatible with HLA-B downregulation by HIV.
Hepatitis C virus (HCV)
Individuals exposed to HCV can either clear infection or remain chronically infected. Those that remain chronically infected have a substantial risk of end-stage liver disease including liver cirrhosis and hepatocellular carcinoma (68). It is now clear that this is a common chronic viral infection affecting more than 1% of the USA population and over 170 million individuals worldwide (69). The dichotomous outcome following infection can be readily determined using a test for HCV RNA. Those individuals who have antibodies to HCV and remain HCV RNA-positive are chronically infected, whereas those that have antibodies without detectable RNA are deemed to have cleared the virus. Comparison of KIR and HLA genotypes from these two groups of individuals have demonstrated a protective association of the inhibitory receptor KIR2DL3 in combination with HLA-CAsn80 (47); however, this protection was only afforded for individuals who were homozygous for both genes and also those with a presumed low inoculum of infection. This sets up a quantitative model for protection in HCV whereby individuals who are homozygous for both KIR2DL3 and HLA-CAsn80 have quantitatively more NK cells that are inhibited by the protective KIR2DL3: HLA-CAsn80 receptor:ligand combination than do individuals who are heterozygous for KIR2DL2/KIR2DL3. Owing to the fact that ~99% of individuals are KIR2DL1-positive, then individuals who are heterozygous for HLA-CAsn80/HLA-CLys80 have a pool of NK cells inhibited by KIR2DL1: HLA-CLys80, which subtracts from the pool of protective NK cells inhibited by KIR2DL3:HLA-CAsn80. This quantitative model is consistent with murine CMV infection in which a quantitative effect of virus inoculum over NK mediated resistance to infection is observed (70). Also, these results suggest that the protective effect more likely arises from expression of KIR on NK cells than on T cells. Furthermore, the expression of inhibitory receptors for HLA class I is a fundamental mechanism for NK cell biology, but not for T cells. A question arises as to why KIR2DL3 is protective, but KIR2DL2, which has the same HLA-C ligands, is not. Binding data suggest that KIR2DL3 binds more weakly to HLA-C than does KIR2DL2 (71), implying that the KIR2DL3:HLA-CAsn80 interaction is more easily perturbed than that of KIR2DL2:HLA-CAsn80 perhaps leading to more effective activation and viral control. Conversely, direct affinity measurements have been made for both KIR with HLA-C ligands and have been found to be similar (72, 73). No direct “head-to-head” comparison of the two proteins has been made to date, however, and this may be crucial as affinity for HLA-C can be dramatically altered both by the specific HLA allele studied and also the specific peptide presented by the HLA molecule. Additionally, the different KIR:HLA-C interactions may transmit signals of different strengths in addition to, or instead of, differences in binding (74). Thus, like many genetic associations, much more functional and biochemical work is required to tease out the true relevance of these associations.
Cytomegalovirus (CMV)
The observation that NK cell–deficient individuals are susceptible to herpes virus infections and that NK cells are crucial for a rapid and effective immune response to murine CMV infection, suggests that NK cells are important for clearance of human CMV infection (8, 9). Nevertheless, although this virus has many mechanisms for influencing MHC class I expression, no specific KIR has been shown to be important in the immune response to CMV (75). This may reflect a true absence of effect or, alternatively, the complexity of the interactions of CMV, MHC class I, and the NK cell repertoire. Instead, there are quantitatively more NK cells expressing NKG2C present in CMV-exposed individuals, suggesting that this NK cell receptor is of particular relevance in the immune response to this infection (76). Furthermore, a single individual whose NK cells all expressed KIR2DL1 developed recurrent infections including CMV, suggesting that expression of this inhibitory receptor may overcome the benefit afforded by NKG2C (77).
Other Pathogens
Malaria
The effect of KIR on the immune response to pathogens extend beyond viral infections. NK cells are important in the immune response to malaria as they can interact with red blood cells infected with the malaria parasite and liberate the pro-inflammatory cytokine interferon-γ (78). In a study of twenty-three individuals, an association between the KIR3DL2*002 allele and the NK cell response to infected red blood cells was found. The molecular basis for this relationship––and whether this KIR allele is acting as a marker for another disease-associated gene––needs further investigation, as red blood cells are deficient in HLA class I, the ligand for KIR3DL2 (79).
Pregnancy
NK cells are found in abundance in the deciduas of the pregnant uterus, early in gestation and coincident with the implantation of the fetal trophoblast (80). They have thus been implicated in the process of “placentation,” in which the placenta grows, invades the uterine decidua, and develops a blood supply adequate to nourish the developing fetus. Inadequate placentation is one mechanism that leads to pre-eclampsia, a state hallmarked by the clinical appearance of raised blood pressure and protein in the urine that if allowed to progress can lead to eclampsia, characterized by the development of epileptic seizures and subsequent fetal and maternal death (81). Adequate placentation is therefore important in reproduction, and it is thought that NK cells may have an important role in this process.
The trophoblast expresses the HLA class I ligands for NK cell receptors HLA-C, -E and -G, rather than HLA-A and -B, indicative of its potential to regulate maternal NK cell functions (82, 83). Study of the maternal KIR and fetal HLA types has suggested that these genes may be risk factors for the development of pre-eclampsia (84). The KIR gene locus can be grouped into haplotypes designated A and B where haplotype A, the most common haplotype, expresses KIR genes KIR2DL1, 2DL3, 2DL4, 3DL1, 3DL2 and a single activating receptor KIR2DS4. The group B haplotypes essentially consist of all other KIR combinations, but importantly, contain more activating receptors. In the pre-eclampsia study, haplotype A was weakly associated with development of disease, however this association was strengthened in the context of the fetus having at least one HLA-CLys80 allele. The KIR2DL1:HLA-CLys80 interaction is considered to be one of stronger inhibition. Furthermore, an inverse correlation was observed between the numbers of activating KIR genes present in the mother and the prevelance of pre-eclampsia. Together these data suggest that strong inhibitory signals and weak activating signals are associated with pre-eclampsia. The proposed mechanism is that the maternal NK cells are overly inhibited by the fetal trophoblast, thus preventing adequate invasion of the decidua and leading to inadequate placentation with inadequate development of the spiral arteries, leading to pre-eclampsia. Evidence suggests that this process has been driving the natural selection of the KIR and HLA-C genes because a population analysis indicates an inverse correlation between the frequency of KIR AA haplotypes and that of HLA-CLys80. Overall, therefore, this is a model that appears similar to that for hepatitis C in that it is based on the hypothesis that KIR2DL1:HLA-CLys80 transmits a stronger inhibitory signal than KIR2DL3:HLA-CAsn80. Confirmation of these genetic studies with functional experiments is now required. Intriguingly, one other group has suggested that if there are too many inhibitory receptors, then spontaneous abortion may result (85, 86). It is, therefore, interesting to speculate that the KIR gene locus has been shaped by a combination of reproductive success and pathogen defense.
Autoimmune Disease
Rheumatoid arthritis
A number of autoimmune disorders have been associated with specific KIR genes and a common theme of these studies is that specific activating KIR are implicated in disease pathogenesis. The first of these was rheumatoid arthritis, in which KIR2DS2 was found to be a factor for the development of rheumatoid vasculitis (87), probably owing to the expression of KIR2DS2 on a subset of T-cells which are CD4+CD28−, and cytotoxic in nature (88). This T-cell subset is expanded in rheumatoid arthritis and was originally found to express the activating NK receptor KIR2DS2 in the absence of corresponding inhibitory receptors (89). Expansion of these auto-reactive T-cell clones can be driven by the KIR:MHC class I interaction in addition to that of the TCR:MHC interaction. Furthermore, the subset of these KIR-positive T cells that express the adaptor molecule killer cell–activating receptor-associated protein (KARAP)/DAP-12, which is required for optimal signaling by activating KIR, can be activated directly through KIR2DS2 to release cytokines and become cytotoxic, without the need for conventional TCR stimulation by antigenic peptides (90). This provides a further mechanism for driving an inflammatory reaction. Immunogenetic analysis, however, did not reveal an association of KIR with rheumatoid arthritis per se, but rather implicated KIR2DS2 (an activating KIR) with rheumatoid vasculitis in a subgroup of individuals with rheumatoid arthritis. These studies illustrate the finding that although KIR are expressed predominantly on NK cells they may have additional abilities to modulate T cells and, thus, directly drive the adaptive immune response. Interestingly, a further correlation at a functional level has been observed in individuals with acute coronary syndromes in which the same phenotype of T cells––CD4+CD28–KIR2DS2+––are more commonly found in affected individuals than in healthy controls (91). Thus there appears to be some consistency in the observations in the relationship of KIR to vascular disease.
Psoriasis and other disorders
Studies of KIR genes in other autoimmune disease, in contrast to rheumatoid arthritis, do not have functional correlates and therefore the associations observed may be due to an influence on NK cells or on T cells. In these circumstances the NK cells could be enhancing the adaptive immune system to break tolerance or the T cells expressing KIR have a primary pathogenic role.
Psoriasis vulgaris is an inflammatory skin disorder that had been associated with the HLA class I molecule Cw6. This HLA class I allele is one of the HLA-CLys80 group and is a ligand for KIR2DS1 (activating) and KIR2DL1 (inhibitory). Genetic studies in both European and Japanese populations have demonstrated an increased frequency of KIR2DS1, both alone and in combination with HLA-Cw6, in individuals with this disease compared to controls (92, 93). Psoriatic arthritis is a similar condition that shares the HLA-Cw6 association and has additional genetic components that may modulate the disease phenotype. A large study of individuals with Psoriatic arthropathy (PsA) suggests that this disease is associated with KIR and HLA class I haplotypes that predispose to activating receptor:ligand interactions (94). In this model, those individuals who have an activating KIR, either KIR2DS1 or KIR2DS2, and are homozygous for either C group 1 or C group 2 HLA-C ligand are most susceptible to PsA, whereas those with no activating KIR (and therefore, have more limited potential for inhibition conferred through inhibitory KIR) are relatively protected from disease. At a genetic level, a hierarchy can be established in which genotypes that would predict a phenotype with more activating receptor:ligand interactions is most susceptible; those with no activating receptor: ligand interactions and most inhibitory receptor:ligand interactions most protected and those with an intermediate number of activating and also inhibitory interactions relatively neutral. This interesting model awaits formal evaluation in other autoimmune disorders especially those in which studies have observed correlations with activating receptors. For instance, a weak association of an activating receptor:ligand pair (KIR2DS2:HLA-CAsn80) has also been observed in diabetes mellitus (95). Additionally, in the autoimmune condition scleroderma, KIR2DS2, in the absence of its inhibitory counterpart KIR2DL2, is a disease susceptibility marker (96). In this study, however, no correlation with the HLA-C ligands for these molecules was performed and, also, individuals lacking KIR2DL2 frequently have KIR2DL3, which has the same HLA-C ligand specificity as KIR2DS2 and KIR2DL3. Collating the data from these genetic studies seems to indicate that the KIR genotype of an individual can furnish that individual with an NK cell repertoire that leans away from inhibition and more towards activation. Such an individual may be relatively protected from viruses, cancer and pre-eclampsia, but susceptible to autoimmunity (Figure 3⇓); however, these genetic studies are relatively recent and require confirmation at a functional level.
Genetic model for the association of KIR with different disease. The activating and inhibitory KIR of different KIR locus haplotypes generate a “molecular rheostat” in which individuals are susceptible or protected from a variety of diseases. This model is based on KIR genotypes and KIR:HLA combinations which are considered to provide different levels of inhibition and activation, and on disease studies in which an association with protection or susceptibility was associated with specific KIR genes, or specific KIR and HLA combinations.
Hematological Disease
The role of KIR in hematological disease extends to that of diagnosis, disease pathogenesis, prognosis and therapeutics and encompasses both genetic and functional studies. The main areas of study have been in relatively small studies of hematological malignancies and larger studies of bone marrow transplantation.
Large Granular Lymphocytic Leukemia (LGL)
In this disease, the KIR may act as a marker of clonality and potentially may be involved in disease pathogenesis. Leukemias of large granular lymphocytes may represent clonal proliferations of either cytotoxic T cells (T-LGL) or Natural Killer cells (NK-LGL) (97). The former is the more common, although both may present with similar clinical manifestations of cytopenias, neuropathies, recurrent infections, splenomegaly, or rheumatoid arthritis (98). Most individuals are asymptomatic and the clinical burden is not predicted by current phenotypic markers.
T-LGL is typified by a common phenotype associated with terminally differentiated effector memory cytotoxic T cells, namely CD45RA+CD27−CD28−CCR7−. In health, this subpopulation of T cells expresses KIR and, in one study, 48% of cases of T-LGL expressed a single KIR, as determined by antibodies to CD158a (either KIR2DL1 or KIR2DS1), CD158b (either KIR2DL2, KIR2DL3, or KIR2DS2), and CD158e (KIR3DL1), supporting the notion of clonal expansion in T-LGL (99). Furthermore, Nowakowski and colleagues recently reviewed seven patients with T-LGL who demonstrated KIR expression of the same genes. In five of seven individuals there was no HLA class I ligand present for the expressed KIR (100). All five cases exhibited splenomegaly and cytopenia and required treatment. Conversely, the two cases with a matched KIR-MHC class I allotype were asymptomatic. This may be relevant to pathogenesis, as was suggested for a case of pure red cell aplasia in the context of KIR-positive T-LGL (101). Interestingly, expression of LILRB1 (also termed ILT2/LIR-1) another inhibitory receptor from the Leukocyte Receptor Complex with a broad HLA class I specificity, was found on T-LGL from asymptomatic patients, suggesting that this receptor may be preventing autoreactivity in these patients (102).
In NK-LGL, KIR were present in 36% of cases and were clonally expressed. The physiological relevance of this finding could not be further interpreted as an analysis of the MHC class I was not undertaken. Zambello and colleagues demonstrated homogeneous KIR-specific mAb staining in eleven of eighteen NK-LGL cases. Thirteen of the cases were genotyped and none were homozygous for the “inhibitory” group A haplotype, with all having at least one of the activating KIR commonly found on the B groups of haplotypes. Furthermore, functional data was collected in seven cases and all exhibited “activating” KIR function (103). These observations are similar to those findings by Epling-Burnette and colleagues (104) in a further thirteen patients whereby the T cells of these patients showed a similar activating KIR expression, providing the tentative speculation that both T and NK cell expansion is driven by the activating KIR.
Cutaneous T Cell Lymphoma (CTCL)
Cutaneous T cell lymphomas are a heterogeneous group of lymphomas that primarily involve the skin. They include Mycosis Fungoides (MF), Sezary syndrome (SS) and CD30+/− pleomorphic T-cell lymphoma. Recently, Bagot and colleagues described the selective expression of KIR3DL2 (CD158k/p140) on CD4+ malignant cells of Sezary syndrome patients (105, 106). Moreover, Wechsler and colleagues showed that CD158k could distinguish in situ CD4+ clonal cells in patients with SS from those with MF (107). Thus, although the role of this inhibitory receptor in this disease is currently unclear, it could still become a valuable diagnostic marker.
Acute Myeloid Leukemia (AML)
There are limited studies of KIR in the pathogenesis of this disease, which appears more closely linked to alterations in the functions of the natural cytotoxicity receptors (108); however, one study found an association between KIR2DL2 and KIR2DS2 and the development of leukemia. This analysis was refined to demonstrate an increased frequency of a specific combination of KIR, which included all the inhibitory receptors and only two activating receptors KIR2DS2 and KIR2DS4, in patients with AML as compared to healthy controls (109).
Solid Tumors
In murine models, NK cells determine the rejection of a wide variety of tumors, and the decreased expression of MHC class I in many tumors also implicates NK cells in the human anti-tumor immune response. The impact of KIR on the development of solid tumors, however, is much less well-documented. At a genetic level, KIR can influence malignancies associated with viral infections. For example, cervical neoplasia is driven by the human papilloma virus in a sequence thought to progress from inflammation to dysplasia to cancer. In a study of >800 women, individuals with cervical neoplasia were more likely to have KIR and HLA genotypes associated with activation. Thus, individuals considered to have the most and the predicted strongest inhibitory KIR:HLA receptor ligand interactions (HLA-CLys80 homozygotes and HLA-Bw4) were most protected from disease whereas those with the fewest, predicted weakest, interactions (HLA-CAsn80 homozygotes and HLA-Bw4−), and a haplotype that included the activating receptor KIR3DS1, were most susceptible to this disease. In this system, KIR3DS1 may be acting as a marker of other disease associated genes, but the model suggests that a more “activating” haplotype predisposes the patient to inflammation following viral infection and subsequently cancer.
A contrasting finding is present in hepatocellular carcinoma (HCC) that follows chronic hepatitis C (HCV) infection. When 150 patients from a single centre were studied for HLA and KIR genotype, a synergistic association was seen with KIR3DS1 in combination with HLA-Bw4Ile80 allotypes and protection against the development of HCC (110). A weak protective effect of KIR3DS1 and HLA-Bw4 against chronic HCV infection was also found in the study of Khakoo et al. (47). This information, if confirmed in multi-center studies, would provide stratification of risk groups for HCC development. In contrast to the associations seen in virally driven cancers, only weak statistical associations were seen in malignant melanoma patients compared to age- and sex-matched controls (111). In the melanoma study, there appeared to be a marginal influence of HLA-C genotype in the presence of disease, in that individuals homozygous for HLA-CLys80––and, by implication, the KIR2DL1: HLA-C pairing––were relatively protected.
Studies of tumors and their infiltrating lymphocytes have yielded little clarification on these matters. NK cells can infiltrate into HCC and these NK cells had lower amounts of KIR and NKG2A expression than did the NK cells from healthy livers (112), suggesting that the low KIR- and NKG2A-expressing NK cells might participate by controlling tumor growth. In colorectal tumors, however, despite their low levels of HLA class I, NK cells are relatively sparse but T cells are present (113). Furthermore, expression of inhibitory KIR on tumor-infiltrating T lymphocytes may also modulate their functions and prevent them from killing cancerous cells (114, 115). Also the shedding of soluble ligands for activating MHC class I receptors on NK and T cells, such as MIC-A and -B, can block the interaction of these cells with tumors thereby modulating the balance between activation and inhibition of NK cells that express normal receptor levels (116, 117). Thus, the complexity of these data reflect the large variety of potential pathogenic mechanisms, immune responses and “immune escape” mechanisms seen in cancers of different origins.
NK Therapy: Current and Future Opportunities
The genetic diversity of KIR and the range of diseases in which they are thought to have specific effects would suggest that they have the potential to form good therapeutic targets. To date, the utility of KIR as a target for intervention has come from studies of bone marrow transplantation.
In the specialized context of HLA-mismatched stem-cell transplantation, a beneficial effect of NK cells upon a variety of leukemias has been observed (118). In these individuals, donor-derived NK cells proliferated after transplantation and targeted myeloid leukemias. A favorable outcome for AML was then demonstrated when there was KIR-MHC class I incompatability in the Graft versus Host (GvH) direction (119). In haplo-identical transplantation, only one MHC haplotype is shared between donor and recipient. Thus, alloreactivity exists when the donor possesses a KIR receptor whose cognate MHC class I ligand (i.e., C1, C2, or Bw4) is not expressed by the recipient. In this case, the recipient will not have a ligand for the inhibitory receptors expressed on a sub-population of donor derived NK cells and these cells will be alloreactive in the GvH direction. This GvH alloreactivity can be directed against the tumor (Graft versus Leukemia (GvL) effect, and in the above study, the probability of leukemia relapse was 0% in the NK mismatch group as compared to 75% in the non-mismatched group. Five year survival was 60% in the mismatched group and 5% in the non-mismatched group. Such retrospective observations were supported by murine adoptive therapy and autologous AML transfer studies where eradication of tumor could be shown only when NK cell alloreactivity was present. Most recently, studies of stem cell transplants for AML in which the donor and recipient were siblings with identical HLA class I types, have shown an improved outcome when there is a donor inhibitory KIR mismatch or when the donor possesses the two activating KIR: 2DS1 and 2DS2 (120, 121).
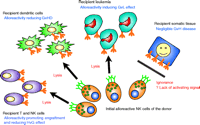
In addition to the beneficial effects of NK mismatching upon GvL, there appears to be a favorable effect of such a mismatch upon engraftment and graft versus host disease (GvHD). Asai and colleagues identified the suppression of GvHD by activated natural killer cells in mouse models after allogenic bone marrow transplantation (122). Ruggeri and colleagues extended these observations within their retrospective study of haploidentical AML transplantation. Although 13.7% of patients experienced GvHD in the absence of a KIR mismatch, no GvHD was seen when a NK mismatch existed in a GvH direction. With regard to engraftment, no rejection was seen in the mismatched group whereas 15.5% rejected the transplant when no mismatch was present (119). Therefore, in contrast to T cell alloreactivity, NK cell alloreactivity appears to both mediate a successful GvH for certain leukemias and prevent GvHD (Figure 4⇓). The ability to prevent GvHD was observed in mouse models when preconditioning with mismatched NK cells allowed the transfer of large numbers of potentially alloreactive T cells, which would normally lead to lethal GvHD. The mechanism of this role of NK cells may relate to the susceptibility of host dendritic cells to NK cell mediated lysis, as GvHD can be prevented by inactivation of host dendritic cells (123). Furthermore, NK cells can proliferate upon interaction with donor dendritic cells and kill such cells through a Nkp30-dependent pathway (4). Although these effects illustrate an important biological point, the protective model of NK cell alloreactivity appears confined to bone marrow transplantation for myeloid malignancies in the context of severe T-cell depletion regimens (124–126).
Potential mechanism for the protective effect of haploidentical bone marrow transplantation in acute myeloid leukemia. A subpopulation of donor derived NK cells do not express an inhibitory receptor for recipient HLA class I. These NK cells are able to lyse recipient T and NK cells (preventing rejection), recipient dendritic cells (preventing rejection), and recipient leukemic cells (preventing relapse). Conversely, these NK cells do not seem to attack somatic tissues.
The exploitation of NK alloreactivity may become an important therapeutic strategy in the management of myeloid malignancy and in the modulation of engraftment procedures and GvHD control. Manipulation of the inhibitory KIR, activating KIR, natural cytotoxicity receptors (NCR; NK receptors that are involved in anti-tumor activity), and lectin family of NK receptors may allow for the selective expansion and administration of NK cells for the future management GvL and GvHD in human studies.
Miller et al. have made infusions of NK cells derived from haplo-identical donors for AML, and also for solid tumors i.e., melanoma and renal cell carcinoma (127). AML patients were treated with high dose chemotherapeutic regimens, whereas patients with solid tumors were treated with less intense chemotherapy; a significant expansion of “alloreactive” NK cells were noted only in the AML patients. This study is consistent with the findings in bone marrow transplantation that the transfer of potentially reactive NK cell clones appears to be well tolerated; however, it also highlights the difficulties in transferring these types of therapies to individuals with solid tumors. Furthermore, NK cells may not gain adequate access to malignant tumors and the tumor may shed soluble molecules that block NK cell activating receptors (117) or promote inhibition (128).
Future developments in the field of NK-mediated therapy of the above diseases are awaited. The genetic models suggest that a successful virological response, but a harmful autoimmune response is associated with a balance towards more activation. Modulation of the key receptor ligand:interactions may have potential for benefit; however, inhibitory signals can be overridden by activating receptor: ligand interactions from receptors other than those of the KIR family, implying that KIR do not necessarily need to be targeted in order to modulate a disease in which they play a role. Rather, the NK cell as a whole should be considered. For instance, Interferon-α, a cytokine used in the treatment of renal cell carcinoma and chronic hepatitis C infection, can augment NK cell cytotoxicity and increases the expression of the activating receptor NKG2D on NK cells (129). In vitro modulation of NK cells by gene transduction using a chimeric molecule is a first step in the process (130); however, until the complex genetic models of these diseases are supported resolutely by functional data the KIR biologist will remain at the bench rather than by the bedside.
- © American Society for Pharmacology and Experimental Theraputics 2005
References

Salim Khakoo, MD, is a Senior Lecturer and Honorary Consultant in Hepatology at Southampton University School of Medicine. His research is focused on natural killer cells and KIR biology, especially the mechanisms by which NK cells counteract viral infections. His clinical interest is in viral hepatitis and its immunopathogenesis. Address correspondence to SK. E-mail sik{at}soton.ac.uk; fax 0 11 44 02380 795152.

Andrew Bateman, MD, PhD, is a Cancer Research UK–funded Clinician Scientist and Honorary Consultant in Radiotherapy and Oncology at Southampton School of Medicine. His clinical interest is in the radiotherapy of the gastrointestinal tract. His research interests include the immunotherapy of solid tumors, the response of natural killer cells to tumors and the interaction of natural killer cells with the adaptive immune system. E-mail arb{at}soton.ac.uk.

Tony Williams, MD, PhD, is a Wellcome Trust Clinician Scientist and Honorary Consultant in Clinical Immunology at Southampton University School of Medicine. His research interests include the cellular biology of MHC class I, antigen processing and presentation, and clinical manifestations of natural killer cell deficiencies. E-mail apw2{at}soton.ac.uk.