|
 
 |
| ORIGINAL ARTICLE |
|
| Year : 2015 | Volume
: 1
| Issue : 1 | Page : 16-19 |
|
Methicillin-resistant Staphylococcus Aureus Nasal Carriage Among Health-care Workers: Decolonization and Follow-up Study Conducted in a Tertiary Care Hospital
P Thilakavathy, GS Vijaykumar, A Ramesh, Anand B Janagond, T Rajendran, SS Jeremiah, G Vithiya
Department of Microbiology, Velammal Medical College Hospital and Research Institute, Anupanadi, Madurai, Tamil Nadu, India
| Date of Web Publication | 10-Aug-2015 |
Correspondence Address:
Dr. P Thilakavathy
Department of Microbiology, Velammal Medical College Hospital and Research Institute, Anupanadi, Madurai - 625 009, Tamil Nadu
India
 Source of Support: None, Conflict of Interest: None  | Check |
 
Background: Hospitals worldwide are increasingly concerned by methicillin-resistant Staphylococcus aureus (MRSA). In recent years, nasal colonization of MRSA among health-care workers (HCWs) is one of the major sources of nosocomial infections in the hospital. The aim of the study was to find out the prevalence of MRSA carriers among the heath care workers and antimicrobial susceptibility pattern of the isolates and postdecolonization screening for clearance of MRSA. Materials and Methods: Nasal swabs of HCWs were collected, cultured, and identified as MRSA using standard microbiological procedures. Antibiotic susceptibility was done by Kirby-Bauer disk diffusion method according to Clinical and Laboratory Standards Institute guidelines. Decolonization with mupirocin ointment was given to MRSA carriers and postdecolonization screening was done. Results: Nasal carriage rate among the HCW was found to be 3.25%. MRSA isolates showed higher resistance erythromycin (40%) followed by ciprofloxacin (30%) and all the isolates were sensitive to amikacin, teicoplanin, linezolid, mupirocin, and rifampicin. Successful decolonization was done for 9 (2.9%) MRSA carriers. Conclusion: Screening and decolonization of HCWs MRSA status should always be part of a comprehensive infection control policy including staff education and emphasizing high compliance with hand hygiene and contact precautions. Keywords: Decolonization, methicillin-resistant Staphylococcus aureus, mupirocin
How to cite this article:
Thilakavathy P, Vijaykumar G S, Ramesh A, Janagond AB, Rajendran T, Jeremiah S S, Vithiya G. Methicillin-resistant Staphylococcus Aureus Nasal Carriage Among Health-care Workers: Decolonization and Follow-up Study Conducted in a Tertiary Care Hospital. J Hum Health 2015;1:16-9 |
How to cite this URL:
Thilakavathy P, Vijaykumar G S, Ramesh A, Janagond AB, Rajendran T, Jeremiah S S, Vithiya G. Methicillin-resistant Staphylococcus Aureus Nasal Carriage Among Health-care Workers: Decolonization and Follow-up Study Conducted in a Tertiary Care Hospital. J Hum Health [serial online] 2015 [cited 2018 Aug 21];1:16-9. Available from: http://www.jhhjournal.org/text.asp?2015/1/1/16/162524 |
| Introduction | |  |
Staphylococci are widespread in nature, their normal habitats being the skin and mucous membranes of human beings. Human skin is densely colonized with several of the coagulase-negative species, and to a lesser extent with Staphylococccus aureus (S. aureus).[1] The anterior nares of nose are the most frequent carriage site for S. aureus. Extra nasal carriage sites that typically harbours S. aureus include the skin, perineum, pharynx and less frequent carriage sites include the gastrointestinal tract, vagina, and axilllae. [2],[3],[4]
Staphylococcus aureus is one of the most prevalent and clinically significant pathogens, causing wide variety of infections ranging from mild skin and soft-tissue infections to serious life-threatening infections. [5] Multi-drug resistant strains of S. aureus have been reported with increasing frequency worldwide. Methicillin-resistant S. aureus (MRSA) infections account for 40-60% of all nosocomial S. aureus infections in many centers across the world. MRSA is a problem in hospitals worldwide and an important cause of health-care-associated infections since 1970's. [6] Since health-care workers (HCWs) are at the interface between hospitals, long-term care facilities, and nursing homes on the one hand and the community on the other, they may serve as reservoirs, vectors, or victims of MRSA cross-transmission. The present study was conducted to find out the prevalence of MRSA carriers among the heath care workers and antimicrobial susceptibility pattern of the isolates and followed up after decolonization with mupirocin ointment.
| Materials and Methods | |  |
The study was carried out from March 2014 to July 2014 in Velammal Medical College Hospital and Research Institute. After obtaining ethical clearance, a total of 308 HCWs were enrolled in the study. The study group included doctors, nurses, midwives, technicians, patient care executives, and written informed consent was obtained from all HCW, who were included in the study. Nasal swabs were collected from each HCW.
Sample collection
Sterile cotton swabs were used for sample collection. The sample was obtained by rotating the swabs gently for five times on both nares of the study participants so that the tip is entirely at the nasal ostium level, and it was transported to the laboratory.
Bacterial isolates
Of the 308 nasal swab samples, 48 were identified as S. aureus. The isolates from samples were identified and characterized using standard microbiological procedures. Swabs were plated on Mannitol Salt Agar (MSA) and incubated at 37°C. After overnight incubation, the plates were examined, and coagulase test (slide and tube) was performed. Those isolates that produced yellow colored colonies on MSA and coagulase positive were taken as S. aureus. The S. aureus isolates were subjected to antibiotic susceptibility test by Kirby-Bauer's disk diffusion method on Mueller-Hinton (MH) agar and the zones were interpreted as per Clinical and Laboratory Standards Institute guidelines. [7]
The following antibiotic disks were used cefoxitin (CXT 30 μg), ciprofloxacin (CIP 5 μg), erythromycin (E 15 μg), clindamycin (CD 15 μg), pristniamycin (RP 15 μg), teicoplanin (TEI 30 μg), mupirocin (MU), and rifampicin (RF 5 μg).
Identification of methicillin-resistant Staphylococcus aureus
For the bacterial isolates that were confirmed to be S. aureus, inoculums were prepared by making direct broth suspension of isolated colonies selected from 18 to 24 h agar plate. The suspension was adjusted to achieve a turbidity equivalent to 0.5 McFarland turbidity standard and inoculated on MH agar. Cefoxitin disk of strength 30 μg was placed on the plates and incubated for 24 h at 35°C. Colonies with an inhibition zone 22 mm were read as MRSA (ATCC 25923) used as control strain.
Methicillin-resistant Staphylococcus aureus decolonization treatment
For MRSA nasal carriers, mupirocin 2% ointment is advised to apply twice daily for 5 days.
Postdecolonization screening for clearance
Nasal swabs were obtained 1-week and 12 weeks after completing decolonization treatment to ensure clearance of the organism. [8]
Statistical analysis
The findings were statistically analyzed Chi-square test (χ2 ) and P value (P < 0.05, statistical significant).
| Results | |  |
A total of 308 HCWs were involved. The distribution of S. aureus and MRSA nasal carriage among different groups of HCWs are shown in [Table 1]. MRSA nasal carriage was high among staff nurse (1.95%) followed by doctors (0.65%), technicians (0.32%) and housekeeping (0.32%).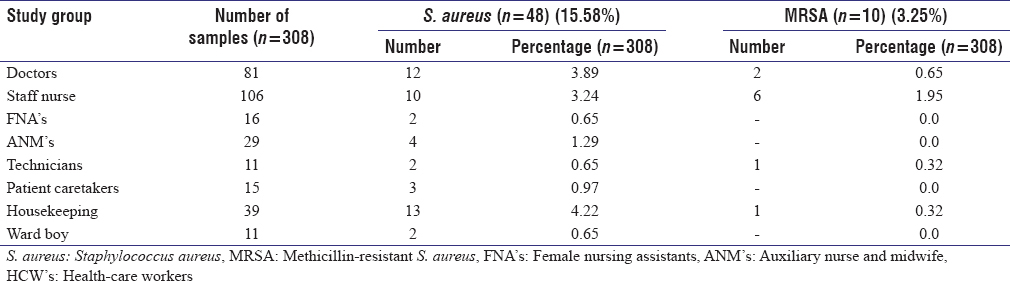 | Table 1: Distribution of S. aureus and MRSA nasal carriage rate among different group of HCW's
Click here to view |
The distribution of S. aureus and MRSA colonization among different wards/OP/OT is shown in [Table 2]. MRSA nasal carriage rate was high in wards (14%) and OP (6%).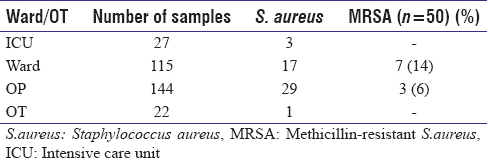 | Table 2: Hospital wards/OP/OT wise distribution of S. aureus and MRSA isolates
Click here to view |
The risk factors and its association with MRSA colonization are shown in [Table 3]. The close contact with infected wound patients was one of the major risk factors associated with MRSA colonization. None of the other risk factors such as associated infections, antibiotic usage, and were significantly associated with MRSA colonization.
Based on the resistance to cefoxitin, MRSA was identified. MRSA isolates showed varying sensitivity pattern. The highest resistance was noted against erythromycin (40%) followed by ciprofloxacin (30%), pristinamycin (10%), and gentamicin (10%). All the isolates were sensitive to amikacin, teicoplanin, linezolid, mupirocin, and rifampicin [Table 4].
| Discussion | |  |
In the present study, S. aureus nasal carriage rate was 15.6% and MRSA nasal carriage rate was 3.2%. Similar findings were noted in the study conducted by Goyal et al. [9] The prevalence rate of MRSA nasal carriage among HCWs was relatively less compared to other studies. [10],[11],[12] These differences could be explained by local prevalence of MRSA as well as infection control measures followed by HCWs.
The nasal carriage rate varied among different professions. MRSA nasal carriage rate was high among staff nurses (1.95%) particularly those who are working in wards (14%) and close contact with infected wound patient was found to one of the risk factors closely associated with MRSA colonization. This could possibly be explained by the high frequency of patient contact among these professionals as reported in a study conducted by Shibabaw et al. [13]
Methicillin-resistant S. aureus isolates showed higher resistance erythromycin (40%) followed by ciprofloxacin (30%) and all the isolates were sensitive to amikacin, teicoplanin, linezolid, mupirocin, and rifampicin. Similar findings were noted in a study conducted by Al-Zaidi. [14] Mupirocin resistance had been reported from South India, but in the present study all the MRSA isolates were sensitive to mupirocin. [15],[16]
In some studies, MRSA colonized HCWs were allowed to work without restrictions with strict emphasis on hand hygiene and standard precautions which had been followed in the present study and also all the MRSA carriers were from wards and none of them from Intensive Care Unit. But in some studies the HCWs were allowed to work only in dedicated MRSA areas or where MRSA was present. [17],[18] It had been reported that the colonized or infected HCWs were temporarily removed from patient care for varying durations until documentation of negative follow-up cultures was obtained. [19],[20]
In the present study, after application of mupirocin ointment, postdecolonization screening for MRSA clearance was done. In our study, 9 carriers (2.9%) were follow-up cultures were negative after 1-week and 12 weeks and documented as successfully decolonized and one of the HCWs did not take part in the further study.
| Conclusion | |  |
The study has been conducted in a newly established hospital in which the study population comprised of study subjects from various backgrounds. Findings of this study indicate that early screening and decolonization of MRSA carriers can reduce the MRSA prevalence rate in hospitals. Screening of HCW for MRSA carriage can be included as part of preemployment examination.
Acknowledgment
Velammal Medical College Hospital and Research Institute, Anupanadi.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
| References | |  |
| 1. | Collee JG, Fraser AG, Marmion BP, Simmons A, Mackie and McCartney. Practical Medical Microbiology. 14 th ed. Churchill Livingstone, Elsevier. New Delhi, 2006. p. 245-6.  |
| 2. | Williams RE. Healthy carriage of Staphylococcus aureus: Its prevalence and importance. Bacteriol Rev 1963;27:56-71.  |
| 3. | Ridley M. Perineal carriage of Staph. aureus. Br Med J 1959;1:270-3.  |
| 4. | Guinan ME, Dan BB, Guidotti RJ, Reingold AL, Schmid GP, Bettoli EJ, et al. Vaginal colonization with Staphylococcus aureus in healthy women: A review of four studies. Ann Intern Med 1982;96:944-7.  |
| 5. | Lowy FD. Staphylococcus aureus infections. N Engl J Med 1998;339:520-32.  |
| 6. | Fluit AC, Wielders CL, Verhoef J, Schmitz FJ. Epidemiology and susceptibility of 3,051 Staphylococcus aureus isolates from 25 university hospitals participating in the European SENTRY study. J Clin Microbiol 2001;39:3727-32.  |
| 7. | Clinical and Laboratory Standards Institute. Perfomance Standards for Antimicrobial Susceptibility Testing: CLSI Document M100-S22.Wayne, Pennysylvania, USA, 2012. p. 32  |
| 8. | |
| 9. | Goyal R, Das S, Mathur M. Colonisation of methicillin resistant Staphylococcus aureus among health care workers in a tertiary care hospital of Delhi. Indian J Med Sci 2002;56:321-4.  [ PUBMED]  |
| 10. | Shakya B, Shrestha S, Mitra T. Nasal carriage rate of methicillin resistant Staphylococcus aureus among at National Medical College Teaching Hospital, Birgunj, Nepal. Nepal Med Coll J 2010;12:26-9.  |
| 11. | Vinodhkumaradithyaa A, Uma A, Shirivasan M, Ananthalakshmi I, Nallasivam P, Thirumalaikolundusubramanian P. Nasal carriage of methicillin-resistant staphylococcus aureus among surgical unit staff. Jpn J Infect Dis 2009;62:228-9.  |
| 12. | Mathanraj S, Sujatha S, Sivasangeetha K, Parija SC. Screening for methicillin-resistant staphylococcus aureus carriers among patients and health care workers of a tertiary care hospital in south India. Indian J Med Microbiol 2009;27:62-4.  [ PUBMED]  |
| 13. | Shibabaw A, Abebe T, Mihret A. Nasal carriage rate of methicillin resistant staphylococcus aureus among Dessie Referral Hospital Health Care Workers; Dessie, Northeast Ethiopia. Antimicrob Resist Infect Control 2013;2:25.  |
| 14. | Al-Zaidi JR. Methicillin Resistant Staphylococcus Aureus (MRSA) nasal carriage among health care workers in Intensive Care Units. Med J Babylon 2014;11:3.  |
| 15. | Dardi CK, Narayan PA. Mupirocin resistance in clinical isolates of methicillin-resistant Staphylococcus aureus from a Tertiary Care Rural Hospital. Int J Adv Med Health Res 2014;1:52-6.   |
| 16. | Oommen SK, Appalaraju B, Jinsha K. Mupirocin resistance in clinical isolates of staphylococci in a tertiary care centre in South India. Indian J Med Microbiol 2010;28:372-5.  [ PUBMED]  |
| 17. | Berthelot P, Grattard F, Fascia P, Fichtner C, Moulin M, Lavocat MP, et al. Implication of a healthcare worker with chronic skin disease in the transmission of an epidemic strain of methicillin-resistant Staphylococcus aureus in a pediatric intensive care unit. Infect Control Hosp Epidemiol 2003;24:299-300.  |
| 18. | Saiman L, Cronquist A, Wu F, Zhou J, Rubenstein D, Eisner W, et al. An outbreak of methicillin-resistant Staphylococcus aureus in a neonatal intensive care unit. Infect Control Hosp Epidemiol 2003;24:317-21.  |
| 19. | Cox RA, Conquest C. Strategies for the management of healthcare staff colonized with epidemic methicillin-resistant Staphylococcus aureus. J Hosp Infect 1997;35:117-27.  |
| 20. | Wagenvoort JH, De Brauwer EI, Sijstermans ML, Toenbreker HM. Risk of re-introduction of methicillin-resistant Staphylococcus aureus into the hospital by intrafamilial spread from and to healthcare workers. J Hosp Infect 2005;59:67-8.  |
[Table 1], [Table 2], [Table 3], [Table 4]
|