 |
 |

Selective Serotonin Reuptake Inhibitors
Infrequent Medical Adverse Effects
Richard J. Goldberg, MD
Arch Fam Med. 1998;7:78-84.
ABSTRACT
The selective serotonin reuptake inhibitors have a very favorable safety profile. However, because of the wide use of this drug group, even low-frequency adverse effects become more prevalent. This article reviews 5 of these relatively infrequent adverse medical effects associated with the use of selective serotonin reuptake inhibitors: the syndrome of inappropriate antidiuretic hormone secretion, extrapyramidal effects, bleeding complications, cardiac arrhythmias, and the serotonin syndrome.
INTRODUCTION
The selective serotonin reuptake inhibitors (SSRIs) are among the most widely prescribed drugs in the world. This drug group has achieved such a phenomenal usage rate partly because of a favorable safety and tolerability profile. However, with such large numbers of patients receiving SSRIs, low-frequency medical adverse effects become more prevalent. It is important for clinicians to heighten their awareness of these adverse effects and not to assume that the SSRIs are devoid of potential medical complications. The purpose of this article is to review 5 of the relatively infrequent adverse medical effects associated with the use of SSRIs: syndrome of inappropriate antidiuretic hormone secretion (SIADH), extrapyramidal symptoms (EPS), bleeding complications, cardiac arrhythmias, and the serotonin syndrome.
SYNDROME OF INAPPROPRIATE ANTIDIURETIC HORMONE SECRETION
The syndrome of inappropriate antidiuretic hormone secretion is characterized by a reduced ability to excrete water, resulting in extracellular dilution with resulting hyponatremia. Cases of SIADH associated with SSRIs have been described in conjunction with fluoxetine,1-7 paroxetine,8-9 or sertraline treatment.7, 10-12
Other Psychotropic Drugs Reported to Cause SIADH
Other drugs have been reported to cause SIADH, including carbamazepine,13 tricyclic antidepressants,14-16 monoamine oxidase inhibitors (MAOIs),17 and neuroleptics.14, 18 While haloperidol has been reported to cause SIADH,19 a study of haloperidol's effects on antidiuretic hormone (ADH) secretion in normal subjects found that hormone levels did not change significantly after haloperidol injection.20 The syndrome of inappropriate antidiuretic hormone secretion has also been reported as a component of mood disorders in patients not receiving psychotropic medications.21-22
SSRI Mechanism for Inducing SIADH
It is not known how the SSRIs induce SIADH. It has been suggested that the SSRIs cause the release of ADH.23-24 Another possible explanation is that the SSRIs increase renal responsiveness to ADH.25
Clinical Symptoms
Clinical signs associated with SIADH generally do not occur until the sodium level falls below 130 mmol/L, and many patients do not manifest significant symptoms until the sodium level falls below 125 mmol/L. The levels of hyponatremia in published case reports have ranged from 98 to 126 mmol/L. The onset of SIADH and clinical symptoms may occur as early as 2 days after starting treatment with an SSRI, or may emerge after many months of taking the medication. Symptom severity depends on the degree of hyponatremia. Typical early signs and symptoms include confusion, generalized weakness, decreased appetite, and general malaise. Later signs and symptoms include headache, somnolence, coma, seizures, transient focal neurological signs, and abnormal electroencephalogram results.
Laboratory Symptoms
While patients with SIADH are hyponatremic, they do not manifest edema, hypotension, azotemia, or dehydration. However, because of increased intravascular volume, it is not unusual to find decreases in levels of chloride, blood urea nitrogen, creatinine, and uric acid. Hypokalemia is not typical of SIADH, but has been reported. Serum osmolalities are low, and case reports show values ranging between 242 and 272 mOsm/kg. The urine sodium level is typically elevated (>30 mmol/d), ranging between 46 and 102 mmol/d. Urine osmolarity is greater than serum osmolarity.
Medical Differential Diagnosis
The syndrome of inappropriate antidiuretic hormone is characterized by the excessive secretion of ADH in the absence of normal osmotic or physiologic stimuli (such as hypotension, decreased plasma volume, or increased serum osmolarity). Other medical disorders causing SIADH include malignant tumors (especially lung, duodenum, and pancreas), intracranial diseases, congestive heart failure, hypothyroidism, hepatic cirrhosis, and pulmonary diseases such as pneumonia and emphysema. Several medications are also reported to cause SIADH. When SIADH is associated with thiazide diuretics,26-27 it is generally accompanied by hypokalemia (unlike SIADH associated with SSRIs), alkalosis, and normal or increased levels of serum creatinine and blood urea nitrogen. Other common medications associated with SIADH include carbamazepine, narcotics, nicotine, phenothiazines, and tricyclic antidepressants.
Psychogenic polydipsia should also be considered as a possible cause of hyponatremia. However, because normal kidneys are able to excrete large volumes of water, pure polydipsia (in the absence of SIADH) rarely leads to hyponatremia. The syndrome of inappropriate antidiuretic hormone secretion has been reported as a component of symptoms consisting of acute psychosis and massive water ingestion.28-29 Psychogenic polydipsia has also been reported as a clinical feature that sometimes accompanies schizophrenia and primary affective disorders. Psychogenic polydipsia should not be considered as a primary diagnosis when symptoms occur in the context of SSRI use.
Medical Management
Symptoms secondary to SIADH improve after correction of the electrolyte disturbance through elimination of the causative agent and restriction of fluid intake. Mild forms often clear up with fluid restriction alone. Severe cases may require diuresis using furosemide and electrolyte replacement. In general, serum sodium levels should be corrected only halfway to normal in the initial 24-hour period to avoid complications such as cerebral edema or central pontine demyelination. A typical fluid restriction would be limitation of intake to about 1000 mL/d. The time necessary to return serum sodium levels to normal has ranged from 48 hours to 6 weeks after the discontinuation of fluoxetine treatment. Some patients can tolerate readministration of an SSRI without a recurrence of SIADH; however, 1 case of relapse has been reported.3
Conclusions
Most cases of SIADH associated with SSRIs have involved elderly patients, and there is some question as to whether older patients, for some unknown reason, are more prone to develop this complication. It also appears that older patients take longer to recover. Therefore, it is important to monitor serum sodium levels in elderly patients receiving SSRIs who develop any behavioral symptoms possibly consistent with hyponatremia.
EXTRAPYRAMIDAL SYMPTOMS
The medical literature contains numerous case reports describing the onset of EPS following the initiation of treatment with SSRIs. A variety of EPS have been reported to be associated with the SSRIs, ranging from tremor to dystonic reactions.30-32 For example, Meltzer et al33 reported a severe dystonic reaction in a 25-year-old man on the fourth day of treatment with 30 mg of fluoxetine twice daily.
Epidemiologic studies have suggested that EPS occur in about 1 of 1000 patients treated with SSRIs.34 Coulter and Pillans30 reported on the prevalence of EPS in patients receiving fluoxetine, calculated by examination of the New Zealand Intensive Medications Monitoring Programme, during a 4-year period. There were 15 reports of EPS among 5555 patients receiving fluoxetine, although some of these patients were receiving other psychotropic medications, including neuroleptics. Fisher et al35 have reported on the incidence of adverse effects in a total of 2786 patients receiving either sertraline or fluoxetine. Adverse effects were reported during the first month of administration in a voluntary monitoring program. A total of 4 patients reported "twitches, tics, or spasms." The Committee on Safety of Medicine 36 listed 39 patients who reported EPS associated with the use of paroxetine of 370000 prescriptions.
Atypical neuromuscular effects, which may be extrapyramidal in origin, have also been reported in association with SSRIs. Stuttering has been reported as a motor dysfunction associated with fluoxetine37-38 as well as sertraline.39 Interestingly, sertraline has also been reported to improve stuttering in a patient,40 possibly as a result of lowering associated depression and social anxiety. Ellison and Stanziani41 reported 4 cases of nocturnal bruxism associated with SSRIs, which were improved by the addition of buspirone to the treatment regimen. Although a few case reports have suggested an antidopaminergic effect of buspirone, it is also possible that buspirone increases striatal dopamine synthesis by blocking presynaptic dopamine inhibitory autoreceptors.42
Akathisia
Akathisia consists of a spectrum of symptoms ranging from a mild subjective sensation of anxiety, to a sense of restlessness, to severe sensations such as "crawling out of my skin." Patients may manifest very little objective restlessness, or may be extremely agitated to the point of requiring restraint. It has been observed for many years that SSRIs induce both psychological as well as motor activation. In many cases, such activation is similar to akathisia associated with the use of neuroleptics. Overall, the effects of the SSRIs on dopamine function are complex and not fully understood. Therefore, while akathisia is included in this section on EPS, it is not clear that akathisia, or other syndromes of "activation" associated with SSRIs are related to their effects on dopamine secretion. The activation from akathisia may be so intense that some patients have attempted suicide.43
Akathisia has been reported to occur in the first week of treatment with SSRIs and may occur with the very first dose.44 If one considers a broad spectrum of physical restlessness (including symptoms of agitation, anxiety, and nervousness), akathisia may have an incidence of up to 20% in patients treated with SSRIs.44-45 Lipinski et al46 reported their estimate of incidence to be between 10% and 25%.
Although akathisia has been reported in association with fluoxetine42, 46-50 and sertraline,51-53 no cases have been reported with paroxetine. However, there is no reason to believe it does not occur in association with this medication.
Akathisia can be managed in a number of ways. Since akathisia seems to be a dose-related phenomenon in most cases, the initial strategy is to consider reduction or elimination of the medication that caused the symptoms. If this is not possible, akathisia symptoms can often be reduced or eliminated by the use of low-dose 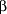 -blockers (such as propranolol in doses of 5-10 mg 3 times daily or clozapine in doses of approximately 0.5 mg 3 times daily).4 Antidepressant-induced mania should also be a diagnostic consideration in patients who experience motor activation after starting treatment with SSRIs. -blockers (such as propranolol in doses of 5-10 mg 3 times daily or clozapine in doses of approximately 0.5 mg 3 times daily).4 Antidepressant-induced mania should also be a diagnostic consideration in patients who experience motor activation after starting treatment with SSRIs.
SSRI Mechanisms for Inducing EPS
Selective serotonin reuptake inhibitors appear to indirectly enhance inhibition of the dopamine system by 2 mechanisms: (1) serotonergic projections inhibit the firing of dopamine cells projecting from the substantia nigra; and (2) serotonergic projections inhibit the synaptic release and synthesis of dopamine in the striatum and cortex.48, 54 Conversely, 5-hydroxytryptamine 2 (5-HT2) antagonists disinhibit the dopamine system and can diminish EPS caused by neuroleptic drugs.55 The amount of dopamine antagonism induced by the SSRIs is usually not sufficient by itself to cause EPS. It has been hypothesized that individuals who develop EPS from the use of an SSRI may actually have preclinical Parkinson disease.34
In addition to secondary effects on the dopamine system, SSRIs may produce EPS through inhibition of the P-4502D6 system, which is responsible for the metabolism of many neuroleptics. Therefore, the coprescribing of SSRIs and neuroleptics may increase neuroleptic plasma levels, causing an increased likelihood of EPS.56-57
Conclusions
There appears to be a low but definite incidence of EPS associated with the use of SSRIs alone (in the range of 1 per every 1000 patients). The emergence of EPS may be facilitated by SSRIs in patients who are simultaneously receiving dopamine receptor–blocking drugs.
BLEEDING COMPLICATIONS
The medical literature contains numerous case reports describing the onset of symptomatic bleeding complications after the initiation of SSRI therapy. However, such complications seem to be exceedingly infrequent. Data from Eli Lilly Co, Indianapolis, Ind, indicate that as of June 30, 1990, for more than 2382000 reported patients receiving fluoxetine, there were only 24 reports of increased bleeding times, coagulation disorder, or coagulation times (a rate of only 1 per 100000 patients). Fisher et al35 have reported on the incidence of adverse effects in a total of 2786 patients receiving either sertraline or fluoxetine. Adverse effects were reported during the first month of administration in a voluntary monitoring program. A total of 4 patients reported "bleeding" as an adverse effect. In a study of the use of sertraline in depression after acute myocardial infarction, Shapiro58 noted that bleeding time was increased in 12 patients, decreased in 4, and unchanged in 2.
Aranth and Lindberg59 reported a case of a 40-year-old woman receiving 60 mg/d of fluoxetine who developed heavy menstrual flow, spontaneous ecchymosis, and splenomegaly, which led to discontinuation of fluoxetine. The ecchymosis faded and the spleen size decreased about 4 days after discontinuing fluoxetine treatment. When she was rechallenged with fluoxetine 1 month later, she again developed severe ecchymosis, which again resolved after discontinuation of fluoxetine. Yaryura-Tobias et al60 reported 8 cases of bleeding after fluoxetine therapy in patients receiving fluoxetine for obsessive-compulsive disorder.
SSRI Mechanisms for Producing Bleeding Complications
More than 99% of whole-body serotonin is stored in platelets.61 Under normal circumstances, platelets release serotonin at the site of vascular tears, leading to platelet aggregation and vasodilation (allowing for clotting without thrombosis). In one study, 12 weeks of fluvoxamine treatment (100-200 mg/d) reduced the platelet serotonin level to 11% of pretreatment levels.62 Since SSRIs block serotonin uptake into platelets,63 it might be expected that SSRIs would potentially impair aggregation and prolong bleeding time. In addition, serotonin-induced platelet aggregation can be blocked by 5-HT2 antagonists, such as ketanserin.
There have also been some concerns about a drug interaction between warfarin and the SSRIs because of reports of bleeding incidents in patients receiving this combination.64 The SSRIs and warfarin do not compete for similar protein-binding sites; therefore, there should not be an increase in warfarin activity based on protein-binding displacement. Another possible explanation of a warfarin-SSRI interaction involves the P-450 system. Fluvoxamine inhibits the P-4501A2 system, which is partly responsible for warfarin metabolism. Therefore, warfarin levels and prothrombin times can be increased when these drugs are coprescribed. It also appears that the P-4502C9 system (which is inhibited by fluoxetine) is partly responsible for warfarin metabolism.65
Laboratory Findings Associated With SSRI-Induced Bleeding
Skop and Brown66 summarized data from 15 cases of SSRI-associated bleeding complications, including ecchymosis, epistaxis, bleeding internal hemorrhoids, menorrhagia, ulcerations with hemorrhage, petechiae, and melena. Analysis of the cases revealed that platelet counts were normal, prothrombin and partial thromboplastin times were normal, prolonged bleeding times were found in several cases, and decreased platelet aggregation in response to adenosine 5'-diphosphate and epinephrine was found in several cases.
Medical Management
When bleeding is considered to be possible SSRI-associated adverse effect, SSRI treatment should be stopped and appropriate psychiatric or hematologic consultation requested. The clinical response to the report or observation of a bleeding complication depends on the severity and significance of the symptoms. There remains no consensus on the relevance of this potential adverse effect for patients undergoing surgery, although discontinuation of SSRIs prior to surgery may be sensible, if appropriate in terms of the patient's psychiatric status.
Conclusions
Overall, given a physiologic basis for SSRI-associated bleeding complications, it is remarkable that so few cases have been reported. The extremely low incidence makes one wonder whether there must be some other rare abnormality co-occurring in patients who develop this complication. Nevertheless, there are groups of patients (including patients undergoing surgery) who are at higher risk of prolonged bleeding and future research should look more specifically at this population.
CARDIAC ARRHYTHMIAS
Given the huge number of patients exposed to SSRIs, there are extremely few reported cases of associated cardiac arrhythmias, and even in these cases there is difficulty defining a causative relationship. In the first 2.5 million patients exposed to fluoxetine, there were reports of 34 cases of atrial arrhythmias, 60 cases of bradycardia, 26 cases of varying degrees of heart block, 54 cases of significant ventricular arrhythmias, 24 cases of congestive heart failure, and 42 cases of other arrhythmias (C. Beasley, Dista Products, Indianapolis, Ind, written communication, 1991).
Several electrocardiographic studies of patients without cardiovascular disease have not demonstrated any significant abnormalities associated with SSRIs.67-71 In a review of 456 patients treated with sertraline (including doses of up to 400 mg/d), there were no demonstrable effects on intraventricular conduction or electrocardiographic intervals. Data from 778 patients (including 119 older than 65 years) did not demonstrate any changes in heart rate or conduction times in patients who were followed up with serial 12-lead electrocardiograms during paroxetine clinical trials (Smithkline Beecham Pharmaceuticals, Philadelphia, Pa, written communication, 1994). In normal persons, fluoxetine may be associated with a small decrease in resting heart rate (3-5 beats per minute), without any conduction changes.72-73
The medical literature contains scattered case reports describing the association of SSRIs with cardiac arrhythmias. Buff et al74 reported a case of atrial fibrillation in an 89-year-old woman 1 day after starting treatment with 20 mg of fluoxetine, as well as a case of atrial fibrillation induced by fluoxetine in an elderly woman with preexisting heart disease.14 Ellison et al75 reported 2 cases of women in their 40s with no abnormal cardiac history who developed bradycardia and syncope while receiving fluoxetine. Feder76 reported a case of fluoxetine-induced bradycardia in a 35-year-old depressed woman who had a syncopal episode, with her pulse rate decreasing to 50/min (from a normal rate of 72/min) after her dose of fluoxetine was raised to 80 mg/d. The bradycardia had not occurred at a lower dose. There have been other observations of an increased incidence of bradycardia.67
SSRI Mechanisms for Inducing Cardiac Arrhythmias
Spier and Frontera77 reported 2 deaths in patients being treated with fluoxetine. These case reports prompted some discussion of whether potential SSRI cardiovascular complications were a result of an arrhythmia or coronary vasoconstriction. Vascular resistance is modulated in a complex fashion by serotonin receptors. In healthy people, serotonin causes vasodilation. For example, in the cerebral arteries, the 5-HT1 receptor causes vasoconstriction, which is why sumatriptan succinate (a 5-HT1 agonist) can be used in the treatment of migraine headaches.78 Since this vasoconstriction can also occur in the coronary arteries, sumatriptan is contraindicated in patients with coronary artery disease. Serotonin also plays an important role in angina.79 While serotonin has a vasodilatory effect on normal coronary arteries (an effect that is blocked by the 5-HT2 receptor antagonist ketanserin),80 serotonin produces direct unopposed vasoconstriction in damaged endothelium. In patients with damaged endothelium (such as those with significant coronary artery disease), it has been speculated that there may be an increase in myocardial ischemia secondary to an increase in vasoconstrictive serotonin in the environment caused by treatment with SSRIs.8, 77, 81-83
Future research should further explore the potential complications of SSRIs in patients with coronary artery or other peripheral vascular disease. Such populations might include trauma patients, patients undergoing surgery and receiving SSRIs, or patients with unstable angina.
SSRI and Cardiovascular Drug Interactions
The SSRIs may contribute to the development of cardiac dysfunction through drug interactions mediated through the P-450 system. Specifically, SSRIs may contribute to bradycardia through impairment of metabolism of 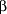 -adrenergic blockers. Fluoxetine has been reported to impair the metabolism of propranolol,84 metoprolol,85 and timolol.86 This effect has not been reported for sertraline.87 The P-4502D6 system is also responsible for the metabolism of type IC antiarrhythmics such as encainide, flecainide, mexiletine, and propafenone,88-90 whose levels may be increased with coprescription of an SSRI. -adrenergic blockers. Fluoxetine has been reported to impair the metabolism of propranolol,84 metoprolol,85 and timolol.86 This effect has not been reported for sertraline.87 The P-4502D6 system is also responsible for the metabolism of type IC antiarrhythmics such as encainide, flecainide, mexiletine, and propafenone,88-90 whose levels may be increased with coprescription of an SSRI.
Conclusions
While there is a high overall level of safety in regard to cardiac effects of the SSRIs, clinicians should consider avoiding SSRIs in patients with bradycardic disorders or tachyarrhythmias and a tendency to atrial fibrillation. Because of P-450 system inhibition, the potential exists for SSRIs to elevate plasma levels of 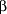 -blockers or type IC antiarrhythmic drugs. -blockers or type IC antiarrhythmic drugs.
SEROTONIN SYNDROME
The increasing use of serotonin-augmenting agents increases the likelihood of "serotonin syndrome."91 Since proper prospective studies have not been conducted, the true incidence is not known. However, there have been reports describing this syndrome, often in association with combinations of medications that include the SSRIs.
Clinical Symptoms
The symptoms that comprise the serotonin syndrome are very common. The array of symptoms that constitute serotonin syndrome include mental status changes, agitation, myoclonus, hyperreflexia, diaphoresis, shivering, tremor, diarrhea, dyscoordination, fever, hypertension, nausea, vomiting, and dizziness. A characteristic cluster of symptoms would include confusion, myoclonus, and gastrointestinal tract activation.
Medications Causing Serotonin Syndrome
The combination of SSRIs with other serotonergic agents, such as lithium (which sensitizes serotonin receptors), buspirone, or tricylics, increases the possibility for this syndrome. The most dramatic cases have been reported as secondary to the combined use of L-tryptophan and MAOIs. However, serotonin syndrome has been associated with several drugs and even more commonly with drug combinations.
It seems unlikely that a patient would develop serotonin syndrome from SSRI treatment alone. However, there is a report of this syndrome following the parenteral administration of citralopam (an SSRI not available in the United States).92 Serotonin syndrome can result from high levels of dextromethorphan (which blocks the neuronal uptake of serotonin).93 Since dextromethorphan is a P-4502D6 substrate, its metabolism is inhibited by the concurrent use of SSRIs. Therefore, this combination of dextromethorphan and an SSRI may predispose to serotonin syndrome.82 The syndrome has been reported in association with a combination of an SSRI with pentazocine,94 MAOIs,95-96 and L-tryptophan.97
A new issue to consider is the potential for serotonin syndrome secondary to the use of SSRIs with weight-loss medications. It has been reported from the combination of fluoxetine and phentermine, an amphetaminelike appetite suppressant with sympathomimetic properties.98 This reaction occurred 8 days after discontinuation of fluoxetine when one 30-mg tablet of phentermine was added to the patient's treatment regimen. The patient developed severe physical and psychological activation with stomach cramps, palpitations, and tremors. This case was reported as an example of sympathetic hyperstimulation resulting from P-4502D6 inhibition of phentermine metabolism, but could also be explained as an example of combined serotonin activity. Dexfenfluramine has recently been approved by the US Food and Drug Administration for the treatment of obesity. It stimulates serotonergic activity by inhibiting serotonin reuptake into presynaptic neurons and enhances serotonin release into brain synapses. Therefore, combining it with an SSRI should be done very cautiously if at all. Because of the long half-life of fluoxetine, it is reasonable to wait about 3 to 4 weeks after its discontinuation before starting dexfenfluramine.
Medical Differential Diagnosis
Serotonin syndrome can be confused with neuroleptic malignant syndrome (NMS),93 since several of the most frequent clinical features overlap. Overlapping features include fever, tachycardia, delirium, diaphoresis, muteness, tremulousness, and abnormal movements. The major clinical feature that distinguishes NMS from serotonin syndrome is the presence of EPS (usually severe rigidity) in NMS as opposed to myoclonus, which occurs in serotonin syndrome. Elevated creatine phosphokinase levels, which are typical for NMS, are not typically present in serotonin syndrome.
The differential diagnosis for serotonin syndrome is similar to that for NMS and includes a variety of central nervous system infections, tumors, central hyperthermia, lupus, and heatstroke. Symptoms of serotonin syndrome can also be produced by a number of drugs including salicylates, anesthetics, anticholinergics, alcohol or sedative withdrawal, toxins (such as carbon monoxide, strychnine, and phenols), and dopamine agonists.
SSRI Mechanism for Inducing Serotonin Syndrome
Normally, serotonin is metabolized by monoamine oxidase A in the liver, or by pulmonary vascular endothelium. The increase in peripheral serotonin caused by SSRI interference with platelet uptake may overcome the body's ability to metabolize peripheral serotonin, leading to a serotonin syndrome. That is why MAOIs (which impair serotonin metabolism), tryptophan (which leads to increased serotonin synthesis), and dextromethorphan (which causes increased release of serotonin) can combine with SSRIs to cause this syndrome.
Medical Management
Symptoms of the serotonin syndrome generally abate within 24 hours after discontinuation of the causative agents. There has been 1 reported case in a patient whose symptoms disappeared after administration of cyproheptadine (a serotonin antagonist).99 It has also been reported that 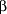 -blockers block 5-HT receptors and inhibit serotonin syndrome caused by L-tryptophan and MAOIs.100 Further, it is notable that propranolol is a 5-HT1 receptor antagonist,101 which is the receptor primarily implicated in serotonin syndrome. Clonazepam can be helpful in relieving myoclonus related to serotonin syndrome. -blockers block 5-HT receptors and inhibit serotonin syndrome caused by L-tryptophan and MAOIs.100 Further, it is notable that propranolol is a 5-HT1 receptor antagonist,101 which is the receptor primarily implicated in serotonin syndrome. Clonazepam can be helpful in relieving myoclonus related to serotonin syndrome.
Conclusions
The widespread use of SSRIs with other serotonergic agents is undoubtedly associated with a spectrum of symptoms of serotonin excess. While the syndrome appears to be self-limited when the offending agent is discontinued, in some cases it can be a life-threatening disorder with rare fatalities reported. In addition, because of the long half-life of fluoxetine's active metabolite, norfluoxetine, patients may be at risk of additive serotonergic reactions for several weeks after discontinuing fluoxetine.
SUMMARY
The use of SSRIs, although associated with a wide margin of medical safety, can result in several rare medical complications. The available data do not make it possible to determine either the incidence or prevalence of these adverse effects because of limitations in study designs and voluntary case-reporting systems. With millions of SSRI users, it is possible that some reported complications are the result of some other concurrent drug or medical disorder independent of the SSRI. The higher numbers of complications associated with fluoxetine are almost certainly because fluoxetine has been marketed for several years before the introduction of sertraline or paroxetine. There is also some possibility that SSRI complications are underreported because the reporting mechanism is too cumbersome for practicing physicians. At this time, it seems reasonable to conclude that the widespread use of the SSRIs results in some serious medical complications of which clinicians should be aware.
AUTHOR INFORMATION
Accepted for publication December 28, 1996.
Corresponding author: Richard J. Goldberg, MD, Department of Psychiatry, Rhode Island Hospital, Providence, RI 02903.
From the Departments of Medicine and Psychiatry, Brown University; and Rhode Island Hospital, Providence, RI.
REFERENCES
1. Viswanath B, Navalgund AA, Cusano W, Navalgund KA. Fluoxetine as a cause of SIADH. Am J Psychiatry. 1991;148:542-543.
WEB OF SCIENCE
| PUBMED
2. Blackstern J, Birt J. Syndrome of inappropriate secretion of antidiuretic hormone secondary to fluoxetine. Ann Pharmacother. 1993;27:273-274.
3. Cohen B, Mahelsky M, Adler L. More cases of SIADH with fluoxetine. Am J Psychiatry. 1990;147:948-949.
4. Kazal L, Hall DL, Miller LG, et al. Fluoxetine-induced SIADH: a geriatric occurrence? J Fam Pract. 1993;36:341-343.
WEB OF SCIENCE
| PUBMED
5. Hwang A, Magraw R. Syndrome of inappropriate secretion of antidiuretic hormone due to fluoxetine. Am J Psychiatry. 1989;146:399.
6. Gommans J, Edwards R. Fluoxetine and hyponatremia. N Engl J Med. 1990;307:103-106.
7. Jackson C, Carson W, Markowitz J, et al. SIADH associated with fluoxetine and sertraline therapy. Am J Psychiatry. 1995;152:809-810.
8. Goddard C, Patton C. Hyponatremia associated with paroxetine. BMJ. 1992;305:1332.
9. Chua TP, Vong SK. Hyponatremia associated with paroxetine. BMJ. 1993;306:143.
10. Crews JR, Potts NL, Schreiber J, et al. Hyponatremia in a patient treated with sertraline. Am J Psychiatry. 1993;150:1564.
11. Thornton S, Resch D. SIADH associated with sertraline therapy. Am J Psychiatry. 1995;152:809.
12. Llorente M, Gorelick M, Silverman M. Sertraline as the cause of inappropriate antidiuretic hormone secretion. J Clin Psychiatry. 1994;22:543-544.
13. Uhde T, Post R. Effects of carbamazepine on serum electrolytes: clinical and theoretical implications. J Clin Psychopharmacol. 1983;3:103-106.
WEB OF SCIENCE
| PUBMED
14. Sandifer M. Hyponatremia due to psychotropic drugs. J Clin Psychiatry. 1983;44:301-303.
WEB OF SCIENCE
| PUBMED
15. Luzecky M. The syndrome of inappropriate secretion of antidiuretic hormone associated with amitriptyline administration. South Med J. 1974;67:495-497.
WEB OF SCIENCE
| PUBMED
16. Abbott R. Hyponatremia due to antidepressant medications. Ann Emerg Med. 1983;12:708-710.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
17. Peterson JC, Pollak RW, Mahoney JJ, et al. Inappropriate antidiuretic hormone secondary to a monamine oxidase inhibitor. JAMA. 1978;239:1422-1423.
FREE FULL TEXT
18. DeRivera J. Inappropriate secretion of antidiuretic hormone from fluphenazine therapy. Ann Intern Med. 1975;82:811-812.
19. Peck P, Shenkman L. Haloperidol-induced syndrome of inappropriate secretion of antidiuretic hormone. Clin Pharmacol Ther. 1979;26:442-444.
WEB OF SCIENCE
| PUBMED
20. Kendler K, Weitzman R, Rubin R. Lack of arginine vasopressin response to central dopamine blockade in normal adults. J Clin Endocrinol Metab. 1978;47:204-207.
FREE FULL TEXT
21. MacMillan H, Gibson J, Steiner M. Hyponatremia and depression. J Nerv Ment Dis. 1990;178:720-722.
22. Zubenko GS, Altesman RI, Cassidy JW, et al. Disturbances of thirst and water homeostasis in patients with affective illness. Am J Psychiatry. 1984;141:436-437.
WEB OF SCIENCE
| PUBMED
23. Kirmura T, Matsui K, Sato T, et al. Mechanism of carbamazepine (Tegretol) induced antidiuresis: evidence for release of antidiuretic hormone and impaired excretion of a water load. J Clin Endocrinol Metab. 1974;38:356-362.
FREE FULL TEXT
24. Brownfield M, Greathouse J, Lorens S. Neuropharmacological characteization of serotoninergic stimulation of vasopressin secretion in conscious rats. Neuroendocrinology. 1988;47:277-283.
WEB OF SCIENCE
| PUBMED
25. Hamburger S, Korprivica B, Ellerbeck E, et al. Thiazide-induced syndrome of inappropriate secretion of antidiuretic hormone: time course of resolution. JAMA. 1981;246:1235-1236.
FREE FULL TEXT
26. Fichman M, Kleeman NC, Vorherr H. Diuretic-induced hyponatremia. Ann Intern Med. 1971;75:853-863.
27. Ashouri O. Severe diuretic-induced hyponatremia in the elderly. Arch Intern Med. 1986;146:1355-1357.
FREE FULL TEXT
28. Dubovsky SL, Grabon S, Berl T, et al. Syndrome of inappropriate secretion of antidiuretic hormone with exacerbated psychosis. Am J Intern Med. 1973;79:551-554.
29. Raskind M, Orenstein H. Acute psychosis, increased water ingestion and inappropriate antidiuretic hormone secretion. Am J Psychiatry. 1975;132:907-910.
WEB OF SCIENCE
| PUBMED
30. Coulter D, Pillans P. Fluoxetine and extrapyramidal side effects. Am J Psychiatry. 1995;152:122-125.
WEB OF SCIENCE
| PUBMED
31. Arya D. Extrapyramidal symptoms with selective serotonin reuptake inhibitors. Br J Psychiatry. 1994;165:728-733.
FREE FULL TEXT
32. Bouchard RH, Pourcher E, Fiteau MS, et al. Dose-related modulation of DA-dependent motor behavior by ritanserin (RIT) in idiopathic Parkinson's disease (PD). Neuropsychopharmacology. 1994;10(suppl):2105.
33. Meltzer H, Younger M, Metz J, et al. Extrapyramidal side effects and increased serum prolactin following fluoxetine, a new antidepressant. J Neural Transm. 1979;45:165-175.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
34. Choo V. Paroxetine and extrapyramidal reactions. Lancet. 1993;341:624.
WEB OF SCIENCE
| PUBMED
35. Fisher S, Thomas KA, Stephen G, et al. Postmarketing surveillance by patient self-monitoring: preliminary data for sertraline versus fluoxetine. J Clin Psychiatry. 1995;56:288-296.
WEB OF SCIENCE
| PUBMED
36. Committee on Safety of Medicine. Dystonia and withdrawal symptoms with paroxetine. Curr Probl Pharmacovigilance. 1993;19:1.
37. Guthrie S, Grunhaus L. Fluoxetine-induced stuttering. J Clin Psychiatry. 1990;51:85.
38. Friedman E. Fluoxetine and stuttering. J Clin Psychiatry. 1990;51:310.
WEB OF SCIENCE
| PUBMED
39. McCall W. Sertraline-induced stuttering. J Clin Psychiatry. 1994;55:316.
40. Costa A, Kroll R. Sertaline in stuttering. J Clin Psychopharmacol. 1995;15:443-444.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
41. Ellison J, Stanziani P. SSRI-associated nocturnal bruxism in four patients. J Clin Psychiatry. 1993;54:432-434.
WEB OF SCIENCE
| PUBMED
42. McMillen BA, Matthews RT, Sanghera MK, et al. Dopamine receptor antagonism by the novel antianxiety drug, buspirone. J Neurosci. 1983;3:733-738.
WEB OF SCIENCE
| PUBMED
43. Rothschild A, Locke CA. Reexposure to fluoxetine after serious suicide attempts by three patients: the role of akathisia. J Clin Psychiatry. 1991;52:491-493.
WEB OF SCIENCE
| PUBMED
44. Boyer W, Feighner J. Side effects of the selective serotonin reuptake inhibitors. In: Boyer W, Feighner J, eds. Selective Serotonin Reuptake Inhibitors. New York, NY: John Wiley & Sons; 1991:133-152.
45. Mendel J. Clinical experience with serotonin reuptake inhibiting antidepressants. J Clin Psychiatry. 1987;48:26-30.
46. Lipinski JF, Mallya G, Zimmerman P, et al. Fluoxetine-induced akathisia: clinical and theoretical implications. J Clin Psychiatry. 1989;50:339-342.
WEB OF SCIENCE
| PUBMED
47. Wirshing W, Van Putten T, Rosenberg J, Marder S, Ames D, Hicks-Gray T. Fluoxetine, akathisia, and suicidality: is there a connection? Arch Gen Psychiatry. 1992;49:580-581.
FREE FULL TEXT
48. Kapur S, Remington G. Serotonin-dopamine interaction and its relevance to schizophrenia. Am J Psychiatry. 1996;153:466-476.
WEB OF SCIENCE
| PUBMED
49. Hamilton M, Opler L. Akathisia, suicidality, and fluoxetine. J Clin Psychiatry. 1992;53:401-406.
WEB OF SCIENCE
| PUBMED
50. Klee B, Kronig M. Case report of probable sertraline-induced akathisia. Am J Psychiatry. 1993;150:986-987.
51. Altshuler LL, Pierre JM, Wirshing WL, et al. Sertraline and akathisia. J Clin Psychopharmacol. 1994;14:278-279.
WEB OF SCIENCE
| PUBMED
52. LaPorta L. Sertraline-induced akathisia. J Clin Psychopharmacol. 1993;13:219-220.
WEB OF SCIENCE
| PUBMED
53. Settle E. Akathisia and sertraline. J Clin Psychiatry. 1993;54:321.
WEB OF SCIENCE
| PUBMED
54. Dewey SI, Smith GS, Logan J, et al. Serotonergic modulation of striatal dopamine measued with positron emission tomography (PET) and in vivo microdialysis. J Neurosci. 1995;15:821-829.
ABSTRACT
55. Ceulemans DL, Gelders YG, Hoppenbrouwers ML, et al. Effect of serotonin antagonism in schizophrenia: a pilot study with steoperone. Psychopharmacology (Berl). 1985;85:329-332.
FULL TEXT
| PUBMED
56. Ciraulo D, Shader R. Fluoxetine drug-drug interactions, I: antidepressants and antipsychotics. J Clin Psychopharmcol. 1990;10:48-50.
WEB OF SCIENCE
| PUBMED
57. Goldberg R. The P-450 system: definition and relavance to the use of antidepressants in medical practice. Arch Fam Med. 1996; 5:406-411.
58. Shapiro P. Treatment of major depression after acute myocardial infarction with sertraline: a preliminary study. Presented at the annual meeting of the American Psychiatric Association; New York, NY; May 1, 1996.
59. Aranth J, Lindberg C. Bleeding, a side effect of fluoxetine. Am J Psychiatry. 1992;149:412.
60. Yaryura-Tobias JA, Kirshen H, Ninan P, et al. Fluoxetine and bleeding in obsessive-compulsive disorder. Am J Psychiatry. 1991;148:949.
61. Artigas F, Sarris MJ, Martinez E, et al. Serotonin in body fluids: characterization of human and cerebrospinal fluid pools by means of a new HPLC method. Life Sci. 1985;37:441-447.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
62. Celada P, Dolera M, Alvqrez E, et al. Effects of acute and chronic treatment with fluvoxamine on extracellular and platelet serotonin in the blood of major depressive patients: relationships to clinical improvement. J Affect Disord. 1992;25:243-249.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
63. Stahl S. Platelets as pharmacologic models for the receptors and biochemistry of monoaminergic neurons in the platelets. In: Longnecker GL, ed. Physiology and Pharmacology. Orlando, Fla: Academic Press; 1985:307-340.
64. Claire R, Servis M, Cram D. Potential interaction between warfarin sodium and fluoxetine. Am J Psychiatry. 1991;148:1604.
65. Goldstein J, deMorais S. Biochemistry and molecular biology of the human CYP2C subfamily. Pharmacogenetics. 1994;4:285-299.
WEB OF SCIENCE
| PUBMED
66. Skop B, Brown T. Potential vascular and bleeding complications of treatment with selective serotonin reuptake inhibitors. Psychosomatics. 1996;37:12-16.
WEB OF SCIENCE
| PUBMED
67. Fisch C, Knoebel S. Electrocardiographic findings in sertraline depression trials. Drug Invest. 1992;4:305-312.
68. Warrington S. Clinical implications of the pharmacology of sertraline. Int Clin Psychopharmacol. 1991;6(suppl):11-21.
69. Edwards J, Goldie A, Papayanni-Papasthatis S. Effect of paroxetine on the electrocardiogram. Psychopharmacology. 1989;97:96-98.
FULL TEXT
| PUBMED
70. Kuhs H, Rudolf G. Cardiovascular effects of paroxetine. Psychopharmacology. 1990;102:379-382.
FULL TEXT
| PUBMED
71. Warrington S, Dana-Haeri J, Sinclair A. Cardiovascular and psychomotor effects of repeated doses of paroxetine: a comparison with amitriptyline and placebo in healthy men. Acta Psychiatr Scand. 1989;80(suppl):42-44.
72. Roose J. Cardiac effects of antidepressant drugs: a comparison of the tricyclic antidepressants and fluvoxamine. Br J Clin Pharmacol. 1983;15:4395-4455.
73. Fisch C. Effect of fluoxetine on the electrocardiogram. J Clin Psychiatry. 1985;46:42-44.
WEB OF SCIENCE
| PUBMED
74. Buff DD, Brenner R, Kirtane SS, et al. Dysrhythmia associated with fluoxetine treatment in an elderly patient with cardiac disease. J Clin Psychiatry. 1991;52:174-176.
WEB OF SCIENCE
| PUBMED
75. Ellison J, Milofsky J, Ely E. Fluoxetine-induced bradycardia and syncope in two patients. J Clin Psychiatry. 1990;51:385-386.
WEB OF SCIENCE
| PUBMED
76. Feder R. Bradycardia and syncope induced by fluoxetine. J Clin Psychiatry. 1991;52:139.
77. Spier S, Frontera M. Unexpected death in depressed medical inpatients treated with fluoxetine. J Clin Psychiatry. 1991;52:377-382.
WEB OF SCIENCE
| PUBMED
78. Hillis W, MacIntyre P. Sumatriptan and chest pain. Lancet. 1993;341:1564-1565.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
79. Golino P, Piscione F, Benedict CR, et al. Local effect of serotonin released during coronary angioplasty. N Engl J Med. 1994;330:523-528.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
80. Golino P, Piscione F, Willerson J. Divergent effects of serotonin on coronary artery dimensions and blood flow in patients with coronary athero-sclerosis and control patients. N Engl J Med. 1991;324:641-648.
WEB OF SCIENCE
| PUBMED
81. McFadden EP, Clarke JG, Davies GJ, et al. Effect of intracoronary serotonin on coronary vessels in patients with stable angina and patients with variant angina. N Engl J Med. 1991;324:648-654.
WEB OF SCIENCE
| PUBMED
82. Skop BP, Finklestein JA, Mareth TR, et al. The serotonin syndrome associated with paroxetine, an over-the-counter cold remedy, and vascular disease: a case report and review. Am J Emerg Med. 1994;12:642-644.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
83. Gardner SF, Rutherford WF, Munger MA. Drug-induced supraventricular tachycardia: a case report of fluoxetine. Ann Emerg Med. 1991;120:194-197.
FULL TEXT
84. Drake W, Gordon G. Heart block in a patient on propranolol and fluoxetine. Lancet. 1994;343:425-426.
85. Walley T. Interaction of metoprolol and fluoxetine. Lancet. 1993;341:967-968.
86. Hayes L, Stewart C, Kim I, et al. Timolol side effects and inadvertent overdosing. J Am Geriatr Soc. 1989;37:261-262.
WEB OF SCIENCE
| PUBMED
87. Ziegler M, Wilner K. Sertraline does not alter the 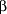 -adrenergic blocking activity of atenolol in healthy male volunteers. J Clin Psychiatry. 1996;57(suppl):12-15.
88. Funck-Brentano C, Thomad G, Jacqz-Algrain E, et al. Polymorphism of dextromethorphan metabolism: relationships between phenotype, genotype and response to the administration of encainide in humans. J Pharmacol Exp Ther. 1992;263:780.
FREE FULL TEXT
89. Crewe H, Lennard M, Tucker G. The effect of selective serotonin uptake inhibitors on cytochrome P-4502D6 activity in human liver microsomes. Br J Clin Pharmacol. 1992;34:262-265.
WEB OF SCIENCE
| PUBMED
90. Lee J, Kroemer H, Silberstein D, et al. The role of genetically determined polymorphic drug metabolism in the beta-blockade produced by propafenone. N Engl J Med. 1990;307:1764-1768.
91. Sternbach H. The serotonin syndrome. Am J Psychiatry. 1991;148:705-713.
WEB OF SCIENCE
| PUBMED
92. Fischer P. Serotonin syndrome in the elderly after antidepressive monotherapy. J Clin Psychopharmacol. 1995;15:440-442.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
93. Kline SS, Mauro LS, Scala-Barnette DM, et al. Serotonin syndrome versus neruoleptic maligant syndrome as a cause of death. Clin Pharmacol. 1989;8:510-514.
WEB OF SCIENCE
| PUBMED
94. Hansen T, Dieter K, Keepers G. Interaction of fluoxetine and pentazocine. Am J Psychiatry. 1990;147:949-950.
WEB OF SCIENCE
| PUBMED
95. Feighner J, Boyer W, Tyler D. Adverse consequences of fluoxetine-MAOI combination therapy. J Clin Psychiatry. 1990;51:222-225.
WEB OF SCIENCE
| PUBMED
96. Sternbach H. Danger of MAOI therapy after fluoxetine withdrawal. Lancet. 1988;2:850-851.
97. Steiner W, Fontaine R. Toxic reaction following the combined administration of fluoxetine and L-tryptophan: five case reports. Biol Psychiatry. 1986;21:1067-1071.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
98. Bostwick JM, Brown TM. A toxic reaction from combining fluoxetine and phentermine. J Clin Psychopharmacol. 1996;16:189-190.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
99. Goldberg R, Huk M. Serotonin syndrome from trazodone and buspirone. Psychosomatics. 1992;33:235-236.
WEB OF SCIENCE
| PUBMED
100. Deakin J, Green A. The effects of putative 5-hydroxytryptamine antagonists on the behaviour produced by administration of tranylcypromine and L-tryptophan or tranylcypromine and L-DOPA to rats. Br J Pharmacol. 1978;64:201-209.
WEB OF SCIENCE
| PUBMED
101. Sprouse J, Aghajanian G. Propranolol blocks the inhibition of serotonergic dorsal raphe cell fring by 5-HT1A selective agonists. Eur J Pharmacol. 1986;128:295-298.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED -adrenergic blocking activity of atenolol in healthy male volunteers. J Clin Psychiatry. 1996;57(suppl):12-15.
88. Funck-Brentano C, Thomad G, Jacqz-Algrain E, et al. Polymorphism of dextromethorphan metabolism: relationships between phenotype, genotype and response to the administration of encainide in humans. J Pharmacol Exp Ther. 1992;263:780.
FREE FULL TEXT
89. Crewe H, Lennard M, Tucker G. The effect of selective serotonin uptake inhibitors on cytochrome P-4502D6 activity in human liver microsomes. Br J Clin Pharmacol. 1992;34:262-265.
WEB OF SCIENCE
| PUBMED
90. Lee J, Kroemer H, Silberstein D, et al. The role of genetically determined polymorphic drug metabolism in the beta-blockade produced by propafenone. N Engl J Med. 1990;307:1764-1768.
91. Sternbach H. The serotonin syndrome. Am J Psychiatry. 1991;148:705-713.
WEB OF SCIENCE
| PUBMED
92. Fischer P. Serotonin syndrome in the elderly after antidepressive monotherapy. J Clin Psychopharmacol. 1995;15:440-442.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
93. Kline SS, Mauro LS, Scala-Barnette DM, et al. Serotonin syndrome versus neruoleptic maligant syndrome as a cause of death. Clin Pharmacol. 1989;8:510-514.
WEB OF SCIENCE
| PUBMED
94. Hansen T, Dieter K, Keepers G. Interaction of fluoxetine and pentazocine. Am J Psychiatry. 1990;147:949-950.
WEB OF SCIENCE
| PUBMED
95. Feighner J, Boyer W, Tyler D. Adverse consequences of fluoxetine-MAOI combination therapy. J Clin Psychiatry. 1990;51:222-225.
WEB OF SCIENCE
| PUBMED
96. Sternbach H. Danger of MAOI therapy after fluoxetine withdrawal. Lancet. 1988;2:850-851.
97. Steiner W, Fontaine R. Toxic reaction following the combined administration of fluoxetine and L-tryptophan: five case reports. Biol Psychiatry. 1986;21:1067-1071.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
98. Bostwick JM, Brown TM. A toxic reaction from combining fluoxetine and phentermine. J Clin Psychopharmacol. 1996;16:189-190.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
99. Goldberg R, Huk M. Serotonin syndrome from trazodone and buspirone. Psychosomatics. 1992;33:235-236.
WEB OF SCIENCE
| PUBMED
100. Deakin J, Green A. The effects of putative 5-hydroxytryptamine antagonists on the behaviour produced by administration of tranylcypromine and L-tryptophan or tranylcypromine and L-DOPA to rats. Br J Pharmacol. 1978;64:201-209.
WEB OF SCIENCE
| PUBMED
101. Sprouse J, Aghajanian G. Propranolol blocks the inhibition of serotonergic dorsal raphe cell fring by 5-HT1A selective agonists. Eur J Pharmacol. 1986;128:295-298.
FULL TEXT
|
WEB OF SCIENCE
| PUBMED
THIS ARTICLE HAS BEEN CITED BY OTHER ARTICLES
Anaesthetic implications of psychoactive drugs
Peck et al.
Contin Educ Anaesth Crit Care Pain 2010;10:177-181.
FULL TEXT
Successful management of depression with reboxetine in a patient who developed parkinsonism related to paroxetine use
Kuloglu et al.
J Psychopharmacol 2010;24:623-624.
Case Report of SIADH Associated With Escitalopram Use
Koski et al.
Journal of Pharmacy Practice 2009;22:594-599.
ABSTRACT
Fluoxetine effect on kidney water reabsorption
Moyses et al.
Nephrol Dial Transplant 2008;23:1173-1178.
ABSTRACT
| FULL TEXT
Antidepressant-Induced Adverse Reactions in a Patient with Hemorrhagic Stroke
Bogdanovic et al.
The Annals of Pharmacotherapy 2005;39:1755-1757.
ABSTRACT
| FULL TEXT
Serotonin syndrome: a brief review
Birmes et al.
CMAJ 2003;168:1439-1442.
FULL TEXT
Adverse Effects of Selective Serotonin Reuptake Inhibitors
Goldberg
Arch Fam Med 1999;8:196-197.
FULL TEXT
|