The Courage of One’s Convictions: THE DUE DILIGENCE OF FRANCES OLDHAM KELSEY AT THE FDA
Half a century has passed since one woman’s steadfastness saved the United States from a disaster that ravaged Germany and a number of other countries. The thalidomide story, including the part played by Frances Oldham Kelsey, has been outlined for readers of Molecular Interventions in an excellent recent article on antiangiogenic drugs (1). Still, it is useful to look at the event more fully. It is a cautionary tale which bears retelling so that we do not repeat the evils of the past. It is also essential that new generations of scientists, who may be unfamiliar with it, become aware of the story and its lessons. The “near miss” of thalidomide may also be compared with a disaster not averted in the US, the Elixir Sulfanilamide tragedy of 1937. The two events led to a strengthening of the American drug laws and the granting of additional powers to the FDA.
These Reflections will delve into Kelsey’s background to show how her research history prepared her for her crucial role in the events of 1960 1962. The supporting role played by another remarkable female physician, Helen Brooke Taussig, will also be brought to the fore.
An Ideal Sleeping Pill
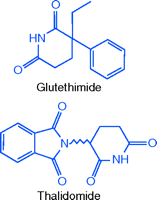
A structural analog of the older sedative glutethimide, the sedative-hypnotic thalidomide (Contergan) was developed by the German pharmaceutical firm Chemie Grunenthal. Toxicity studies in mice, rats, guinea pigs, and rabbits showed it to be remarkably safe, and it was introduced as an over-the-counter (OTC) remedy in West Germany in 1956 (2). By 1960, thalidomide was a top-selling drug in West Germany. At this time, thalidomide was also marketed throughout Europe, in Canada, and in other parts of the world. It produced quick onset, natural sleep, with no hangover; in addition, it relieved the morning sickness of pregnancy. Unlike the barbiturates, it was non-addictive and was too non-toxic to be used in suicide attempts (3).
Marketing rights for the US were obtained from Grunenthal by Richardson-Merrell Inc. (RMI), the parent company of both Wm. S. Merrell Co., in Cincinnati, and The National Drug Company, in Philadelphia. Although both of these divisions were involved in dosage-form development for thalidomide (Box 1), Merrell had the responsibility of filing the New Drug Application (NDA) for the drug. The NDA was submitted in September 1960, and quick approval was anticipated. Under the Food and Drug law in effect at that time, an NDA required only proof of safety. There could be no question that the drug was “safe”; the boast was that it was impossible to kill an animal with thalidomide, no matter how much was administered. To review Merrell’s NDA, the FDA assigned Frances Kelsey, who had joined the Agency only the previous month. The idea was to give the “new girl” something easy to start with.
Thalidomide: A Personal Recollection
As a Richardson-Merrell Inc. (RMI) employee in the pharmaceutical research section of The National Drug Company, I had a minor connection with the thalidomide project. It was RMI’s policy regarding new drugs that they acquired to let both their pharma divisions (RMI’s and NDC’s) market them, in competition, under different trade names. They had carried out this policy in the late fifties with diethylpropion, an anorectic agent licensed from a German firm. Merrell and National separately formulated tablets, and marketed diethylpropion as Tenuate (Merrell) and Tepanil (National). In the case of Contergan (thalidomide), Merrell was to do the heavy lifting develop tablet formulas and file the NDA. We at NDC had only to formulate a liquid dosage form for pediatric use.
Thalidomide is water-insoluble, thus a stable suspension had to be prepared. My assistant pharmacist Paul and I formulated a satisfactory suspension. The only thing needed now was an acceptable flavoring. One day Paul took a quantity of the suspension, divided it into perhaps twenty 100mL aliquots, and incorporated a different flavoring into each one. He then proceeded to taste-test the samples. At 5:00 PM, I walked into his laboratory to check on his progress. There was Paul, sitting on his high lab stool, his head on the lab bench, fast asleep. Fortunately, he was awakened easily and was able to drive home safely. Even more fortunately, Paul was not a pregnant woman.
By law, the FDA had sixty days to review and approve the NDA unless some deficiency was found. At the end of sixty days, instead of the expected approval letter, there came a request for more safety data. Each submittal of additional data triggered another sixty-day review period. As 1960 and 1961 slipped away, RMI and Merrell became increasingly frustrated. Numerous phone calls and personal visits were made to Kelsey, and complaints about her nitpicking were made to her superiors at FDA all to no avail (3).
Was the reviewer vacillating, unable to make up her mind? Was it, as some company people privately suggested, a misplaced feminine intuition, or even caprice? To better explain, we need to look at Frances Kelsey’s background, both personal and scientific.
Vancouver to Washington, by Way of Chicago
Born in 1914, on Vancouver Island, in Western Canada, Frances Oldham was interested in science from an early age. She graduated from McGill University in 1934, in the depth of the Great Depression, and found it impossible to get a laboratory job; therefore, she stayed at McGill and earned an MSc in pharmacology in 1935. At that time, Eugene Geiling was setting up a new pharmacology department at the University of Chicago, and Frances wrote to him at her professor’s suggestion.
Her joy at Geiling’s offer of a research assistantship and scholarship in his PhD program was somewhat tempered by his addressing her in his letter as “Dear Mr. Oldham.” When she approached her professor with this question, he told her to accept the offer, write “(Miss)” after her signature, and head for Chicago. Years later, she told an interviewer that she never was able to find out from Geiling whether he had read Francis instead of Frances (3).
In 1937, her second year at the University of Chicago, Frances was drawn into the investigation of the Elixir Sulfanilamide tragedy alluded to above. In 1935, sulfanilamide, the first of the twentieth-century “miracle drugs,” came on the market. For the first time many previously life-threatening systemic bacterial infections became curable. Sulfanilamide came in 0.5-g tablets, one to four of which constituted a dose. There was a need for a liquid dosage form that could be readily administered to small children and to the many people who have difficulty swallowing large tablets.
Sulfanilamide is not sufficiently soluble in water, alcohol, or glycerin the solvents generally used in making pharmaceutical syrups and elixirs to get a full dose into solution. The Massengill Company of Bristol, Tennessee, found an excellent solvent for the drug, one that was sweet as well: diethylene glycol (DEG). Adding some pink coloring and cherry flavoring to the DEG-sulfanilamide solution, Massengill was able to offer an attractive, pleasant-tasting, easily administered product: Elixir Sulfanilamide. The product was an instant success, but very soon reports of deaths started flowing in (4). Ultimately, there were 107 deaths. To get a feel of what each one of these deaths meant, one need only read a letter sent to President Roosevelt by a mother who lost her six-year-old daughter (4): “Tonight, Mr. Roosevelt, that little voice is stilled.... Even the memory of her is mixed with sorrow for we can see her little body tossing to and fro and hear that little voice screaming with pain and it seems as though it would drive me insane.”
Frances Kathleen Oldham Kelsey receiving the President’s Award for Distinguished Federal Civilian Service from President John F. Kennedy, in 1962.
The deaths were caused by Elixir Sulfanilamide, but why was not obvious. Could the cause be a “bad” batch of sulfanilamide, or something in one of the other ingredients? Geiling, among others, was enlisted by the FDA to help determine the answer. His group performed toxicity tests on rats, rabbits, and dogs, of pure sulfanilamide, pure DEG, commercial Elixir Sulfanilamide, and a “synthetic” Elixir Sulfanilamide compounded according to Massengill’s formula. Their results, and those of other laboratories, confirmed that DEG was the toxic agent in Elixir Sulfanilamide (5) (Box 2). As a doctoral student of Geiling, Frances Oldham assisted in carrying out the animal studies.
Diethylene Glycol Toxicity
Oral ingestion of diethylene glycol (DEG) causes gastrointestinal irritation, followed by anuria, renal failure, pancreatitis, hepatitis, and neurologic dysfunction culminating in coma. DEG induces histologic changes in the kidneys, but unlike ethylene glycol, does not manifest oxalate deposition (10, 11).
It is shocking that, although DEG has been known as a poison since 1937, it still finds its way into medicines in various parts of the world. Unscrupulous or ignorant vendors use it as a cheap adulterant for glycerin. As one example, from November 1995 through June 1996, eighty-six Haitian children were diagnosed with acute renal failure. Ten were transferred to the US for intensive care and dialysis; of these, nine survived. Of the seventy-six children remaining in Haiti, only one survived. The poisoning was traced to two Haitian acetaminophen syrups, made with glycerin that contained DEG. The glycerin had been imported into Haiti from another country (11).
The unfortunate chemist who formulated Elixir Sulfanilamide committed suicide. The Massengill firm was punished with the highest fine permitted under the Food and Drug Law of 1906: $26,100.
Under the 1906 Act, it was not punishable to sell a poisonous drug product. The firm was fined for the risible reason that calling the product an elixir was illegal, as it contained no alcohol. Young Frances Oldham learned two lessons: 1) the importance of thorough pharmacological and toxicological study of any potential medicinal product, and 2) the need for adequate legal and regulatory control over a field affecting the life or death of sick people. The tangible result of the disaster was the enactment of an addition (Section 505) to the Federal Food, Drug, and Cosmetic Act of 1938, requiring that before a new drug may be marketed, the sponsor must submit an NDA containing data to prove the safety of the drug product. Proof of safety had to relate to the entire product, including its excipients, not just to the active ingredient (4).
During the war years of the 1940s, Oldham, continuing in Geiling’s laboratory, became involved in the search for a substitute for quinine, screening compounds for antimalarial action and studying quinine itself. She noted that a Texas veterinarian who submitted an amorphous inky substance for testing wrote that he had tried it on his secretary without ill effects and was planning to administer it to cows. The substance proved inactive. In the course of her studies on quinine, she found that, whereas rabbits metabolize the drug rapidly, pregnant rabbits break it down more slowly, and embryonic rabbits are unable to process it at all. She also observed that some drugs pass from the maternal blood stream, through the placental barrier, into the fetus. These research experiences produced in her what Pasteur would have called the “prepared mind.”
Frances Oldham’s name appears during this period, as coauthor of several studies on antimalarial drugs. In a typical publication, on tissue metabolism of quinine, the senior author is pharmacologist Fremont Ellis Kelsey (6). Laboratory collaboration apparently can lead to personal collaboration: Oldham married Kelsey in 1943. She had received her PhD in 1938, and undertook medical studies. While in medical school, she gave birth to two daughters. She was ahead of her time. Today there is nothing unusual about female medical students and residents concurrently becoming mothers but in the 1940s! Kelsey received her MD at Chicago in 1950. In the following decade, Kelsey reviewed articles for JAMA, taught pharmacology at the University of South Dakota, and worked as locum tenens for doctors in small communities in that state. In 1960, she took a position as medical officer at the FDA, and relocated to Washington, DC.
Kelsey Reviews the Thalidomide NDA
As noted above, Kelsey’s first assignment was to review the NDA for Kevadon, Merrell’s trade name for thalidomide. Her superiors thought it would be an easy one to start with. The drug was already on the market all over Europe. It was known to be safe. Kelsey, however, was no rookie at pharmacology. Her experience had given her a rather sophisticated view of drug pharmacology and safety. Upon reviewing the NDA, she had a number of concerns. She felt that the absorption and excretion data were inadequate, and that the chronic toxicity studies were not long enough. She was even unhappy with the drug’s vaunted non-toxicity; obviously, it wouldn’t kill an animal because most of the drug was not being absorbed. Suppose some illness, concomitant drug, or other condition increases the absorption. She requested more information from the sponsoring firm, but did not find what they submitted satisfactory (3).
In December 1960, scanning the British Medical Journal, Kelsey saw a letter from a physician, reporting cases of peripheral neuritis (tingling in the hands and feet) in patients who had taken thalidomide for a long time. This raised another warning bell in her mind; recalling her studies on quinine, she even considered a possible effect on nerve development in the fetus, should the drug pass the placental barrier. She asked the sponsor for follow-up information. But in place of solid data, she got ever more urgent phone calls and visits from Merrell and RMI representatives. They complained bitterly to her superiors, who, to their credit, refused to overrule her (3). In late 1961, thalidomide began to reveal its face.
The Tragedy of Phocomelia
In November 1961, German doctors became aware of a striking increase in the birth of deformed babies. They were born with abnormally short limbs, “flipper-like” arms, or toes sprouting from the hips. This condition, known as phocomelia, is generally very rare and usually affects only one limb.
Other infants had malformed internal organs, or eye and ear defects. At first, it was suggested that this seeming epidemic might be caused by a virus or by radioactive fallout from the nuclear bomb tests of the 1950s. A painstaking review of the records of the unfortunate mothers, along with personal interviews, however, led Widukind Lenz, a German pediatrician, to conclude that thalidomide, taken in the first trimester, was the causative agent. Lenz immediately informed Grunenthal, and ten days later the German authorities removed the drug from the market, despite Grunenthal’s objections (3).
It is estimated that more than 10,000 children, from among forty-six countries, were born with thalidomide-induced defects. There were seventeen such babies in the US, only because Merrell had distributed some 2.5 million tablets to physicians for “investigational” purposes while the NDA was still pending. Of course, as soon as the news broke, the Kevadon NDA was dead in the water; it was formally withdrawn in March 1962. In January of that year, while the thalidomide investigation was roiling Europe, a remarkable American physician, Helen B. Taussig of Johns Hopkins (Box 3), entered the picture.
Helen Brooke Taussig (1898–1986)
Helen Taussig’s investigation of the thalidomide births and her subsequent testimony before Congress were relatively small episodes in her distinguished career. She had earlier gained a reputation for her studies on congenital cardiovascular malformations. She was able to diagnose and differentiate these problems before the advent of cardiac catheterization (and thirty years before echocardiography became available), largely by the use of the stethoscope and the fluoroscope. Together with Alfred Blalock she developed the Taussig-Blalock shunt to treat so-called “blue babies” surgically. This brought a stream of patients and doctors from all over the world to her cardiac clinic at the Johns Hopkins School of Medicine. In addition, she and Blalock traveled widely in the US and Europe to teach their technique to other surgeons.
Born in Cambridge, MA, into a distinguished academic family, Helen Taussig lost her mother to tuberculosis at the age of eleven. She was dyslexic throughout her life, was often considered mentally retarded by her teachers, and learned to read, write, and spell with help from her father. She graduated from University of California in 1921, having been a championship tennis player in college. Her first choice for medical school, Harvard, was closed to women as degree candidates. She studied medicine at Boston University and Johns Hopkins, receiving her MD from Hopkins in 1927. After serving as intern and resident in pediatrics at Hopkins, she was appointed director of the cardiac clinic, and encouraged by Edwards A. Park, head of pediatrics, to concentrate on congenital heart disease. Her entire career was spent at Johns Hopkins.
The Taussig-Blalock procedure was only one of Taussig’s many contributions to pediatric cardiology. Her 1977 textbook, Congenital Malfunctions of the Heart, was for many years the standard text. She published papers in this field for an astonishing sixty years, from 1926 to 1988. She trained a host of residents and fellows, and befriended many of them. It is said that in 1944 there were only seven pediatric cardiologists in the US; by 1963, Taussig had trained sixty Americans and also many international physicians in this field (12).
All this did not come smoothly. Besides dyslexia, Taussig faced the problem of hearing loss in middle age. She had an amplifying stethoscope made for her, and occasionally made a student or resident jump by passing it to him, and having him listen to the crashing sound of a baby’s heart. She did eventually have surgery to improve her hearing.
Unlike Frances Kelsey, Taussig never married. She found her fulfillment in her nephews and nieces, the spouses and children of her colleagues, and the patients whom she had enabled to live and grow to adulthood. Upon retiring in 1963, she settled in a senior community near Philadelphia, so that she could easily get to the museum and library of the College of Physicians in that city. She died in an automobile accident while on her way to vote at a local polling place (13).
Incomprehensibly, academic advancement was slow in coming to Helen Taussig, a full professorship being granted only four years before her retirement. However, her achievements received due recognition elsewhere. In 1945, she received the National Press Club Award, as well as the title Chevalier Legion d’Honneur of France. In 1964, President Lyndon Johnson presented her with the Medal of Freedom, in honor of her work on prevention and treatment of childhood heart disease (12–14).
Dr. Taussig Investigates
Helen Brooke Taussig was already a famous pediatric cardiologist, having earned the gratitude of untold numbers of parents of children born with heart defects. One of her former fellows, Alois Beuren of Gottingen, alerted her late in January 1962 to the outbreak of phocomelia in Germany. She immediately left for West Germany to investigate at first hand, traveling throughout that country except for West Berlin. Her trip was funded by the International Society for Cardiology Foundation, the Heart Association of Maryland, and NIH.
The urgency and human concern motivating Taussig are illustrated by the fact that she first heard of the problem in late January, went overseas immediately, and published her findings by the end of June, 1962, in JAMA (7). Furthermore, her paper is written in the first person, eschewing the dispassionate tone that the passive voice would convey.
Besides extensive conversations with Lenz, Taussig interviewed many other German physicians. Her paper includes a detailed description of phocomelia and photographs of affected infants. It also provides information on the occurrence of phocomelia in England and other countries where thalidomide was in use. (For information on the future of some of the thalidomide babies, see Box 4.)
Adulthood of Children Affected by Thalidomide
Thalidomide caused major deformities in about 10,000 human beings. Many died shortly after birth, but most survived. Internet searches turn up stories of their later lives, three of which are cited below as examples of the triumph of the human spirit.
One “thalidomide baby,” posting on an adoption Web site, in 1998, reports that his arms end just below his malformed elbows and that his legs are also malformed. Unable to adjust to prosthetic hands, he now uses a simple hook for grasping. He graduated from high school, in 1976, and from college, in 1980, with a degree in computer technology. He opened his own business in 1985. Remarkably, he married in 1981, and he and his wife have three children (15).
In 2002, the BBC marked the fortieth birthday, of about 450 survivors, with a documentary, entitled “Thalidomide: Life at 40.” British parents, having in 1962 formed the Thalidomide Society, reported on their mutual support and the importance of their community. Distillers, the British firm that had marketed thalidomide, agreed to pay an annual sum to each affected child. These payments have continued, despite two corporate mergers, and currently total more than two million pounds per year. Surveys have shown that the afflicted persons’ bodies deteriorate more rapidly than those of the general population, resulting in shortened life spans. Still, they struggle to lead lives as normal as possible, given their handicaps. One woman, born with no limbs, yet married and the mother of a child, is quoted as saying, “I am on four committees, I go dancing, I go to the bingo, I go shopping and I go to the pub” (16).
A final example, and one who has gained recognition and fame as a performance artist, is that of the German bass-baritone Thomas Quasthoff. Born in November 1959, Quasthoff, at four feet tall, shows shortening of the long bones in his legs; the long bones in his arms are shortened or absent. Denied admission to a music conservatory because he is unable to play the piano, he studied voice privately, and studied law for three years. Before launching his musical career in 1988, he worked as a radio announcer and did voice-over for TV. In 2006, Quasthoff married the German TV journalist Claudia Schtelsick.
In 1988, Quasthoff won the ARD International Music Competition in Munich. He made his American debut in 1995, singing Bach, and in 1998 sang as a soloist on a Bach Festival recording that won a Grammy award. He branched into opera, at the Salzburg Festival in 2003, and recorded his first jazz album in 2006. Quasthoff has garnered a number of Grammies, as well as the 2009 Herbert von Karajan Music Prize. He works as a full-time voice professor at the Hanns Eisler School of Music in Berlin (17).
Taussig reported that originally, thalidomide was available over the counter, but when cases of polyneuritis appeared in April, 1961, owing to long-term use, the drug was limited to prescription only. Finally, following Lenz’s discovery, it was removed from the market in Germany and other countries (7).
Through her in-person investigations, Taussig made herself the US expert on thalidomide and its teratogenic effects. She later testified on the subject before Congress, urging that “all new drugs which circulate through the blood stream should be screened for their effect on the offspring of pregnant animals” (7). This requirement has become a sine qua non in the preclinical testing of new drug candidates.
Aftermath
Frances Kelsey’s behind-the-scenes role in protecting American mothers was brought to public attention by reporter Morton Mintz in the Washington Post of July 15, 1962. Follow-up articles on Kelsey, thalidomide, and the need for stronger drug laws aroused the public and led to a steam-roller effect, forcing Congress to act. Kelsey and Taussig both gave testimony before Congressional committees, resulting in passage of the Kefauver-Harris amendments, known as the Drug Amendments of 1962, signed into law by President Kennedy on October 10, 1962 (3).
The 1962 Drug Amendments required that an NDA must provide proof-of-effectiveness as well as proof-of-safety analyses. Equally important, they instituted the Investigational New Drug (IND) Application: the sponsor must let FDA review all preclinical safety testing, as well as the protocols for planned clinical trials, before the new drug is administered to any human subjects. This is intended, inter alia, to shut down the practice of distributing drug samples to doctors before approval, for supposed investigational use, but actually as a form of promotion. Requirements for informed consent were instituted, so that untried drugs could not be administered to unsuspecting patients and adverse drug events would be accessible to the FDA.
Kelsey was appointed head of the newly established investigational drug branch of the FDA and had a hand in developing the regulations for putting the 1962 Amendments into effect. The strengthening of FDA was completed with the appointment, in 1966, of James L. Goddard as Commissioner of Food and Drugs. Goddard proved a tough Commissioner, pushing through what he deemed in the interest of the public health without fear or favor of the regulated industry. Goddard enlisted the National Academy of Sciences/National Research Council (NAS/NRC) to undertake a huge task, reviewing the effectiveness of all drugs approved between 1938 and 1962 on the basis of safety only. This project, the Drug Efficacy Study Implementation (DESI) Program, took many years but resulted in the removal from the market of thousands of drug products whose efficacy was not supported by adequate data.
In 1962, Kelsey received the President’s Award for Distinguished Federal Civilian Service, presented to her personally by President John F. Kennedy. Among the other honors bestowed on Kelsey were a gold key from the University of Chicago (1963), induction into the National Women’s Hall of Fame (2000), and a secondary school in Mill Bay, near her birthplace of Cobble Hill, Vancouver, named in her honor. At a greater distance from Cobble Hill, there now exists an asteroid named “6260 Kelsey” (8). As late as 2001, at age 86, she was serving as deputy of scientific and medical affairs at FDA’s Center for Drug Evaluation and Research (CDER).
As the culminating honor, in 2010, the FDA established the “Dr. Frances O. Kelsey Award for Excellence and Courage in Protecting Public Health,” an annual award to an FDA staff member. The first of these awards was presented on September 15, 2010, by Margaret Hamburg, the current commissioner, to Dr. Kelsey herself (9).
- Copyright © 2011
References
Stanley Scheindlin, DSc, holds a BS in pharmacy from Temple University and graduate degrees in pharmaceutical chemistry from Philadelphia College of Pharmacy and Science (now University of Sciences in Philadelphia). His academic research dealt with plant constituents and chemical interactions of vitamins. In his pharmaceutical industry career, he handled new drug formulation developments, and later regulatory affairs, presiding over the filing of about 100 generic new drug applications and two innovative drug applications. Now retired, his activities include volunteer work, consulting, and writing Reflections pieces for this journal. E-mail: stansch{at}verizon.net