
- Institution: Stanford Univ Med Ctr Lane Med Lib/Periodical Dept/Rm L109
- Sign In as Member / Individual
BLOOD-BRAIN BARRIER DRUG TARGETING: THE FUTURE OF BRAIN DRUG DEVELOPMENT
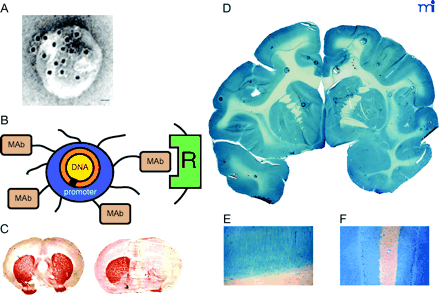
Non-invasive, non-viral gene transfer to the primate brain. A. Transmission electron micrograph of a pegylated immunoliposome (PIL). The IgG molecules tethered to the tips of the 2000-Da polyethyleneglycol (PEG) are bound by a conjugate of the 10 nm gold and an mouse-specific secondary antibody. The position of the gold particles illustrates the relationship of the PEG-extended monoclonal antibody (MAb) and the liposome. Magnification bar = 20 nm. Reprinted with permission (51). B. Plasmid DNA encapsulated in the interior of the PIL, which is conjugated with a receptor (R)-specific targeting MAb. The targeting MAb is conjugated to the tips of 1–2% of the PEG strands that project from the surface of liposome, and there are about 2000 strands of PEG conjugated to the liposome surface. The PEG strands inhibit uptake of the PIL by the reticulo-endothelial system in vivo and enable a prolonged blood residence time of the PIL in vivo (50). The tissue-specific expression of the exogenous gene in vivo can be regulated with the use of tissue-specific promoters incorporated into the plasmid DNA (53). C. Tyrosine hydroxylase (TH) immunocytochemistry of rat brain 3 days after a single intravenous injection of a TH expression plasmid encapsulated in a PIL and targeted to neurons of brain with either a MAb to the BBB TfR (left panel) or a mouse IgG2a isotype control antibody (right panel). Adult rats received an injection of the neurotoxin, 6-hydroxydopamine, on the right side of the brain into the median forebrain bundle 4–5 weeks prior to the intravenous gene therapy. The successful creation of the neurotoxin lesion was confirmed by testing rotation behavior prior to TH gene therapy (51). There is complete normalization of both striatal TH expression and activity ipsilateral to the neurotoxin injection when the TH expression plasmid is effectively delivered to the brain with a TfR Mab–targeted PIL (left panel), because the PIL is able to traverse the BBB via transport on the TfR. However, there is no reconstitution of striatal TH with the control PIL (right panel), because this PIL is unable to cross the BBB. Reprinted with permission (51). D. β-galactosidase histochemistry of brain removed from an adult Rhesus monkey 48 hours after a single intravenous injection of a β-galactosidase-expressing plasmid encapsulated in a PIL conjugated to a human insulin receptor (HIR)-specific MAb. There is global expression of the exogenous gene throughout the primate brain with increased expression in gray matter relative to that in white matter. Panels (E) and (F) are light micrographs of occipital cortex and cerebellum, respectively. The columnar organization of the occipital cortex of the primate brain is revealed (E), and the dense gene expression in the molecular and granular layers of the cerebellum and the intermediate Purkinje cells, are visible in (F). Panels D–F reprinted with permission (49).


