Targeting Lipids to Prevent HIV Infection
The human immunodeficiency virus (HIV) has proven to be a difficult pathogen to overcome; there are currently no effective vaccines that provide specific and long-lasting immunity to the virus. Moreover, there are only a few currently FDA-approved drugs that target HIV proteins such as reverse transcriptase, protease, and the surface envelope protein. Although these drug combination therapies provide effective suppression of HIV virions in individuals, the cost, toxicity, and drug resistance remain common concerns. Until recently, with the FDA approval of Fuzeon® (enfuvirtide), there were no drugs on the market that specifically target and prevent the entry of HIV into human cells. Inhibitors of reverse transcriptase and protease are designed to work only after viral contents have entered into cells, whereas enfuvirtide works by binding to the HIV transmembrane envelope subunit gp41 and preventing the fusion of the viral membrane with the host membrane (1). Other fusion inhibitors involving the blocking of host chemokine receptors are currently being investigated in preclinical models and clinical trials (2). Despite the development of such potential therapeutics, the spread of HIV worldwide continues unabated with no promising cure in sight. Further pursuit of our understanding of HIV will lead to new possibilities for preventing HIV infection that can be effective and affordable. We discuss newly proposed approaches to the prevention of HIV infection by targeting and manipulating host cell lipids, based on recent discoveries regarding the role of lipid rafts in HIV biology.
HIV fusion and infection involves a step-wise process that includes a number of host cell proteins and lipids (3). First, the HIV envelope protein gp120 binds to CD4 on target cells. Many other accessory proteins have also been implicated in mediating HIV binding to host cells as well, including DC-specific inter-cellular adhesion molecule-3 (ICAM-3)-grabbing nonintegrin (DC-SIGN), macrophage mannose receptor, lymphocyte function-associated antigen–1 (LFA-1), intercellular adhesion molecule–1 (ICAM-1), and glycosaminoglycans (4–7). Subsequent to the initial binding to CD4, a conformational change in gp120 allows it to bind to chemokine receptors, generally, either CCR5 or CXCR4, although a number of other chemokine receptors may serve as HIV coreceptors. This binding then triggers a conformational change that exposes a previously buried portion of the transmembrane glycoprotein, gp41, resulting in its insertion into the host cell membrane. The host and viral membranes then fuse, permit-ting the contents of the virus to enter the cell cytoplasm. Recent studies have demonstrated that lipids, such as cholesterol and sphingolipids on both the virus and the host cell, can influence the efficiency of virus-cell fusion (8–11).
Lipid rafts, which are cholesterol- and sphingolipid-enriched membrane domains, appear to be involved in HIV fusion and infection. The formation of lipid rafts arises from the tight packing of cholesterol with saturated sphingolipid chains that allows for increased order in the membrane and resistance to non-ionic detergents at 4°C (12). Studies using β-cyclodextrin (BCD) to remove cholesterol from target cell membranes have demonstrated significant inhibition of HIV infection (10, 11, 13–15). Similarly, chemical inhibition of glycosphingolipid production also resulted in the inhibition of HIV infection (9). Thus, perturbation of lipid raft–specific lipids in the target cell membrane can influence the cell’s ability to be infected. A supposed minor alteration in the cholesterol molecule by oxidation of carbon-3 (to form 4-cholesten-3-one) also results in significant inhibition of HIV infection (16). Although it was initially thought that cholesterol is simply required for membrane mixing, studies have demonstrated that the HIV inhibitory effects of treating cells with BCD can be over-come by over-expressing chemokine receptors (8). Furthermore, BCD treatment alters the conformation of chemokine receptors, which inhibits function as well (14). The CD4 molecule is predominantly localized to lipid rafts under normal physiological conditions; however, the requirement for the interaction of CD4 and lipid rafts in HIV infection is currently being debated: some researchers have demonstrated the interaction to be essential for infection (17), whereas others have found it to be non-essential (18–20). An additional point of interest regarding cholesterol is that statin drugs, originally identified as cholesterol-lowering agents, inhibit HIV infection both by blocking the interaction of ICAM-1 with LFA-1 and by inhibiting Rho GTPase activity (21, 22). Because lipid-altering drugs can influence a number of cell signaling pathways and cell responsiveness, the question “Is the effect a pure membrane inhibitory effect or is it inhibiting cell signaling pathways that may be required for productive infection?” remains to be determined.
Cholesterol in the HIV membrane is also essential for infectivity. As viruses bud from infected cells they do so at cholesterol- rich lipid rafts, resulting in the enrichment of cholesterol in viral membranes as compared to the levels of cholesterol found on host cells (23, 24). Hildreth and colleagues have proposed the use of BCD in topical formulations, which would remove cholesterol from free virions, in order to prevent HIV infection (25). Preclinical studies using such topical compounds are currently underway. Moreover, the HIV Gag protein, which is the major viral structural protein, is modified by the addition of a saturated myristoyl tail promoting its incorporation into lipid rafts. This finding has led others to demonstrate that the addition of unsaturated fatty acids to HIV producing cells can inhibit the production of virions, most likely by covalent modification of Gag, resulting in non-raft protein localization (26).
Although the common approach to manipulating lipid rafts is to target cholesterol in the cell membrane, another approach is to alter raft-associated sphingolipids. In a recent publication, Blumenthal and colleagues have demonstrated that increasing cellular ceramide can inhibit HIV infection (27). Ceramide is a molecule composed of a sphingosine and a fatty acid chain. The addition of a phosphocholine group produces sphingomyelin, a known raft-associated lipid. In their study, the retinoic acid derivative 4-HPR (fenretinide) was utilized to increase cellular ceramide levels in HeLa cells, peripheral blood activated T cells, and monocyte-derived macrophages (MDMs). Such treatment resulted in a significant decrease in the HIV infectivity of the three cell models and with all of the viral strains tested. Some viral strains demonstrated nearly complete inhibition of infection at concentrations of <5 mM 4-HPR, but there was significant variability in the IC50 values and maximal inhibition among the different strains. This HIV inhibition was a ceramide-specific effect, as evidenced through the use of sphingomyelinase, which cleaves sphingomyelin to create ceramide, and by the addition of exogenous ceramide. Unlike BCD, which removes cholesterol from cell membranes, 4-HPR did not affect chemokine receptor function. The increase in cellular ceramide levels inhibited cell-cell fusion, leading to the hypothesis that increasing ceramide affects membrane lipid-raft organization and structure. Based on these data, the authors propose that 4- HPR could potentially be used as an anti-HIV drug in combination with current therapies, based on its low toxicity.
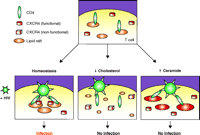
The potential for targeting ceramide raises some interesting aspects regarding HIV therapy. Further studies will need to be done in order to fully characterize how increased ceramide levels may influence the cell membrane. How does 4- HPR and ceramide impact the formation, integrity, and structure of lipid rafts, including signaling protein association and CD4 association? It should be noted that Blumenthal and coworkers also reported that this ceramide-mediated inhibition of HIV infectivity was not observed using VSV-gene-containing pseudotyped or surrogate virions. Given that VSV-pseudotyped viruses do not depend on lipid rafts for entry into cells, these data strongly support a lipid raft–specific but not a whole-membrane effect. Another recent study has demonstrated that ceramide also efficiently displaces cholesterol from lipid rafts (28), thus potentially explaining the raft-specific effect without interfering with CXCR4 cholesterol interactions. Can ceramide somehow prevent the ability to cluster CD4 and chemokine receptors into microdomains that may be required for infection? As there is no known physiologic reason that CD4 would need to interact with chemokine receptors, inhibiting such clustering could potentially be quite specific for HIV infection (Figure 1⇓). For now, as we expect HIV-infected patients to be treated with anti-retrovirals for the rest of their lives, thus, the use of lipid concentration–altering drugs could elicit serious long-term effects, especially in cells such as neurons that are rich in gangliosides and sphingolipids. Do these drugs have immune inhibitory effects, such as with T cell activation? As HIV patients already have suppressed immune systems with decreased CD4 T cell counts, any additional suppression may prove dangerous.
The path to inhibiting HIV infection remains elusive, in which, we will require the development of new approaches to achieve success. Targeting lipid rafts could possibly provide such a means to inhibiting viral infection. Whether BCD formulations in topical microbiocides or 4-HPR will be clinically effective at slowing or stopping HIV infection remains to be demonstrated. At least we can begin to broaden the scope of HIV therapy by understanding how host cell lipids contribute to HIV infection. In addition, the targeting of non-HIV proteins may provide the path of least resistance to achieving effective HIV therapy.
Model for targeting lipid rafts to inhibit HIV infection. On T cells, the HIV receptor CD4 is constitutively associated with lipid rafts at the cell surface, whereas chemokine receptors such as CXCR4 are normally excluded. Upon HIV binding and cell signaling, CXCR4 is recruited to rafts where they can interact with gp120, resulting in viral fusion to the cell membrane (bottom left). Treatment with BCD or statins to remove membrane cholesterol disrupts lipid rafts and cell signaling, resulting in an inhibition of HIV infection and a loss of chemokine receptor function (bottom center). Increasing membrane ceramide levels also alters the properties of lipid rafts (bottom right), possibly by displacing cholesterol from lipid rafts, which results in the inhibition of HIV infection. In contrast to treatment with BCD, increased ceramide in the cell membrane does not result in the loss of chemokine receptor function.
- © American Society for Pharmacology and Experimental Theraputics 2004
References

Dzung Nguyen, PhD, is currently a post-doctoral fellow in the laboratory of Dr. Ajit Varki within the Glycobiology Research and Training Program at the School of Medicine at the University of California, San Diego. E-mail dhn002{at}ucsd.edu Please address correspondence to either DDT or DN.

Dennis D. Taub, PhD, is currently the Chief of the Clinical Immunology Section and the Acting Chief, Laboratory of Immunology at the Gerontology Research Center at the National Institute on Aging, NIH. E-mail Taubd{at}grc.nia.nih.gov; fax 410- 558-8284.