On Cotransmission & Neurotransmitter Phenotype Plasticity
- 1Department of Pharmacology, CNS Research Group, Faculty of Medicine, Université de Montreal, C.P. 6128, Succursale Centre-Ville, Montreal, Quebec, Canada H3C 3J7
- 2Department of Physiology, Neurosciences, and Biophysics, Centro de Investigación y Estudios Avanzados, Apartado Postal 14-740, Mexico D.F. 07000
Abstract
Most neurons in the nervous system appear to contain and release more than one chemical acting as a neurotransmitter or neuromodulator. Cotransmission can therefore be considered the rule rather than the exception. Indeed, cotransmission of a classical neurotransmitter and a peptide is a ubiquitous phenomenon, but several neuron types can also contain more than one classical neurotransmitter [glutamate, γ-amino butyric acid (GABA), acetylcholine, dopamine, etc.]. Although the expression of peptide cotransmitters is known to be highly regulated in response to various physiological, chemical and pathological signals, new data now suggest that a similar situation prevails in neurons that co-release two classical transmitters. In this review we will consider a number of recently described examples of cotransmission implicating more than one classical neurotransmitter. We will also consider new data showing that during development and later in adulthood, as well as in the context of disease, the neurotransmitter phenotype of neurons can be highly plastic as revealed by changes in the expression of neurotransmitter synthesis enzymes and vesicular neurotransmitter transporters.
Introduction
Dale postulated that a neuron functions as a metabolic unit, whereby a process occurring in the cell can influence all of the compartments of that given neuron (1). Through the years, this was unfortunately transformed in the literature to a principle stating that “a single cell releases only one neurotransmitter.” Until recently, this statement has influenced many neuroscientists to consider with skepticism the idea that classical neurotransmitters could be co-released from neurons.
It is now clear that the “one neuron, one neurotransmitter” postulate is the exception rather than the rule. Indeed, the co-release of a fast-acting “classical” neurotransmitter and a peptide, nucleotide (e.g., ATP), Zn2+, neurotrophic factor, nitric oxide, or endogenous cannabinoids—among other modulators—has been established. In all these cases the co-released factors are synthesized in the cell (except for Zn2+), released in a selectively regulated fashion, act on receptors, and are removed or inactivated by specific mechanisms. These characteristics help to confer upon the factors the status of “neurotransmitter,” and they serve a modulatory purpose. On the other hand, the coexistence and co-release of two or more “classical” neurotransmitters, each conveying a “principal message,” has been studied less extensively and has in some ways remained a curiosity. In the present review, our objective is to show that, in fact, co-release is not that uncommon and is now known to exist in a variety of neural systems. In addition, we argue that although many populations of neurons may not release two classical neurotransmitters under basal conditions, many appear to do so early during development or postnatally in response to a variety of physiological and pathophysiological stimuli.
Co-release of Neurotransmitters during Development and in the Adult under Basal Conditions
Co-release of ATP with Acetylcholine or Norepinephrine
One of the first known examples of co-release of two classical or small-molecule neurotransmitters was provided by the discovery that the adenosine-derived nucleotide ATP is co-released with acetylcholine (ACh) from the electric organ of Torpedo (2, 3) and in the neuromuscular junction of the rat (4, 5). ATP appears to be co-released with ACh or with norepinephrine (NE) from neurons of the autonomic nervous system (6–10). The co-release of ATP may serve physiological purposes such as presynaptic inhibition of excitatory neurotransmission secondary to ectoenzyme-mediated catalysis of ATP, and activation of adenosine receptors (11). In pathophysiological conditions (i.e., after neuronal injury), activation of purinergic receptors may also play a neuroprotective role (11).
Co-release in Invertebrate Neurons
Early work in invertebrates has also provided evidence for the co-release of classical neurotransmitters. For example, giant meta-cerebral neurons of Helix aspersa (garden snail) were first identified as using serotonin [5-hydroxytryptamine (5-HT)] as primary neurotransmitter but later shown to also produce and release ACh as a cotransmitter (12). Some 5-HT neurons of Aplysia also contain additional transmitters, including ACh and octopamine (a monoamine closely related in structure to NE) (13), whereas giant neurons from Lymnaea stagnalis (great pond snail) can contain both dopamine and serotonin (14). The specific physiological role of such forms of cotransmission in invertebrate neurons has not been determined.
Co-release of Acetylcholine and Norepinephrine from Sympathetic Neurons
Elegant studies by Patterson and by Furshpan and their colleagues, published in the mid-1970s and early 1980s, demonstrated that postganglionic sympathetic neurons can release ACh as a cotransmitter in addition to NE. These authors placed neurons from the superior cervical ganglion of rats in primary culture together with dissociated heart cells. Recordings from the heart cells showed that the neurons first established contacts releasing NE. Over time in culture, however, the neurons gradually switched to a cholinergic phenotype leading to the production of nicotinic synaptic responses (15, 16). The specific benefits from such a switch are unclear. However, a phenotypic switch of this type apparently recapitulates the development of the sympathetic innervation of sweat glands: outgrowing sympathetic neurons start out with a noradrenergic phenotype but subsequently change to a cholinergic phenotype in response to the release of a secreted factor produced by the sweat glands (sweat gland factor or SGF) (17).
Co-release of GABA and Glycine
GABA and glycine were the first pair of fast-acting neurotransmitters unequivocally proven to be co-released from synapses in the mammalian central nervous system (CNS). This phenomenon was observed in the synapse made by interneurons and motoneurons of the spinal cord of neonatal rats (18). The coexistence of both amino acids in single cells of the spinal cord was already known (19), and it was later determined that these two amino acids are packaged in and released from the same vesicles (20, 21). Interestingly, GABA and glycine are expressed in different amounts and released at different timepoints during development in the lateral superior olive (22) and the medial nucleus of the trapezoid body (23). Of course, although they are co-released, pure GABA type A (GABAA) receptor (GABAAR)- or glycine receptor–mediated synaptic responses can occur as a function of the kind of receptors expressed by different target cells (24). In the spinal cord, fast glycinergic conductances have been proposed to efficiently hyperpolarize the cell, whereas smaller and slower GABAA components could control shunting and the time course of inhibition. Alternatively, differential modulations of GABAA and glycine receptors may permit fast and synapse-specific adaptation of inhibition to network states of activity. Also, the presence of receptors for one or the other kind of neurotransmitter allows for differential modulation and, thus, differential circuit control. This mechanism would be extremely useful, because cell-to-cell signaling is not fully constrained by synaptic wiring and synapse independence (24).
Co-release of Glutamate and GABA
It is noteworthy that a set of GABAergic-glycinergic neurons in the medial nucleus of the trapezoid body can also transiently release glutamate during development (25) (see below). Similarly, GABA is transiently released from the glutamatergic granule cells of the hippocampus (26). Unlike the actions of GABA and glycine, which are both inhibitory amino acids, glutamate and GABA have opposing actions in the adult. In 1991, Sandler and Smith provided the first data to suggest the presence of GABA in the “glutamatergic” mossy fibers of the hippocampal granule cells in monkey and human hippocampi (27). Using GABA-specific antibodies, they found immunoreactive sites in mossy fiber terminals that made asymmetric synaptic contact with spines arising from large dendrites of CA3 pyramidal cells. From this immunoanatomical evidence, they concluded that GABA could either be taken up from the extracellular space or that it originated from an alternative route of synthesis (perhaps from γ-hydroxybutyrate). This latter possibility was posited because neither of the isoforms of the rate-limiting enzyme for GABA synthesis, glutamic acid decarboxylase (GAD), was known to be present in these neurons. Thus, the authors suggested that if the mossy fibers contained GABA, then at least one component of the inhibitory synaptic potentials evoked in pyramidal cells by dentate gyrus stimulation had to be of mossy fiber origin. In this way, GABA released by the mossy fibers could finely modulate the normal mossy fiber glutamatergic responses.
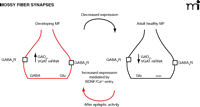
Evidence clarifying the presence and origin of GABA in the mossy fibers was provided by Sloviter et al. These authors conclusively demonstrated that immunoreactive sites specific for GABA and for the 67-kDa isoform of GAD (GAD67) were normally present in the mossy fibers of rats, monkeys, and humans. Seizures increased the amounts of GABA and of both the 65- and 67-kDa isoforms of GAD in mossy fibers (28). Subsequently, electrophysiological methods revealed that the activation of mossy fibers, in the presence of ionotropic glutamate receptor antagonists, could produce GABAAR-mediated monosynaptic responses in CA3 after seizures (29, 30). In developing rodents, the release of GABA from mossy fibers is a normal phenomenon (26, 31, 32); however, this release is transient, because the release of GABA (and the expression of GABAergic markers in the granule cells) ceases upon completion of the development of this pathway (26) (Figure 1⇓). This early GABAergic activity might contribute to the refinement of neuronal connectivity before the establishment of the adult neuronal circuit (33). Other examples in which the expression of a dual glutamatergic-GABAergic phenotype has developmental implications were recently described. For example, the immature GABA-glycinergic projections of the medial nucleus of the trapezoid body transiently express a glutamatergic phenotype (25); this has been proposed as a mechanism for the developmental reorganization of this inhibitory circuit.
Plasticity of the dual glutamatergic-GABAergic phenotype of the granule cells. During development, the granule cells and their terminals express the machinery for the synthesis and release of glutamate and GABA. Upon its completion, at the end of the third postnatal week, the GABAergic phenotype is decreased and the granule cells exclusively express a glutamatergic phenotype. Seizures and sustained enhancement of excitability transiently increase the GABAergic phenotype in adult neurons. MF, mossy fiber.
For a cell to respond to a neurotransmitter, the corresponding receptor must be properly located at the postsynaptic site. Within this context, how can neurons respond to cells releasing two classical neurotransmitters? In cultured neurons, GABAARs and α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptors (AMPARs) can cluster in a single hippocampal pyramidal cell in apposition to different terminals of the same (presynaptic) glutamatergic fiber (34). This evidence suggests that the “mismatching” of the glutamatergic and GABAergic elements on either side of the synaptic connection reflects a common signal involved in the alignment of presynaptic and postsynaptic components during the formation of excitatory and inhibitory synapses (34). Interestingly, excitatory nicotinic receptors and inhibitory glycinergic receptors have also been found apposed to a presynaptic cholinergic terminal, although in distinct postsynaptic membrane microdomains (35). These data suggest that postsynaptic sites possess the ability to adjust to dynamic shifts in the release of neurotransmitters of opposing functions. In particular, in the highly plastic mossy fiber synapse, AMPARs and GABAARs colocalize in apposition to release sites on mossy fibers, where glutamate and GABA coexist (36). Additionally, GABAergic interneurons of the hippocampus contain GABA and aspartate at presynaptic boutons apposed to GABARs and N-methyl D-aspartate (NMDA) receptors (37).
Co-release of Monoamine Neurotransmitters and Glutamate
Although dopamine (DA) and other monoamine neurons in the vertebrate CNS have long been known to contain a number of peptide cotransmitters, such as cholecystokinin (CCK) and neurotensin, recent work now suggests that at least a proportion of such neurons also use glutamate as a cotransmitter (38–41). Immunohistochemical data first revealed that a number of monoamine neurons are strongly immunopositive for glutamate (42–47) as well as for phosphate-activated glutaminase (48), an enzyme involved in the synthesis of neurotransmitter pools of glutamate. Studying isolated 5-HT neurons in culture, Johnson demonstrated that monoamine neurons could, indeed, not only contain but also release glutamate from synaptic vesicles (49). Similar results were subsequently published for DA neurons (47, 50, 51). Finally, more recently, electrophysiological recordings obtained from mouse brain-slice preparations and from anesthetized rats showed that extracellular stimulation of presumed DA neurons in the ventral tegmental area (VTA) generates glutamate-mediated postsynaptic responses in ventral striatal and prefrontal cortex neurons, respectively (52, 53). Earlier indirect results obtained with a slice culture preparation also yielded a similar conclusion (54).
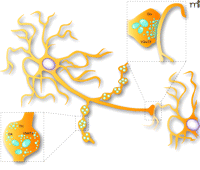
A better understanding of glutamate cotransmission by monoamine neurons requires explaining why these neurons are able to package and release glutamate, whether this is a capacity shared by all mesencephalic DA neurons, and whether all axon terminals of any given glutamate-releasing DA neuron do in fact release both neurotransmitters. Answers to all these questions are not available; however, the recent cloning and characterization of three vesicular glutamate transporters (VGluT1–3) in the CNS has provided important clues (55–67). Interestingly, the initial mapping of the expression of the mRNA of these transporters revealed that VGluT2 is found in subpopulations of noradrenergic neurons in the brain stem whereas VGluT3 is found in 5-HT neurons of the Raphe nuclei (67–69). Finally, although there is no widespread regional expression of any of the three VGluTs in adult mesencephalic DA neurons, VGluT2 is expressed by mesencephalic rat DA neurons in primary culture, as demonstrated by immunocytochemistry and single-cell RT-PCR (70). That a subset of mesencephalic DA neurons in vivo also express VGluT2 was recently confirmed by in situ hybridization (71, 72), validating earlier preliminary reports (73, 74). Further experiments are now required to identify the signals that regulate the expression of VGluT2 and thus of a glutamatergic phenotype by DA neurons in vivo. A current hypothesis is that DA neurons may establish two sets of axon terminals: some that are synaptic and others that are non-synaptic (i.e., free nerve endings) (75). A majority of terminals, most likely those that have been described as non-synaptic could be specialized for the release of DA. The other subset of terminals, showing a more classical synaptic structure with pre- and postsynaptic specialization, could be specialized for the release of glutamate, either alone or together with DA (Figure 2⇓) (38). The physiological significance of dopamine-glutamate cotransmission is also presently unclear; however, DA neurons are known to encode motivational changes, reward predictions or stimulus salience (i.e., novel, infrequent stimuli) via fast transient neuronal activity (76, 77). Considering that DA itself signals through activation of G protein–coupled receptors and cascades that are relatively slow, it is possible that such fast information encoding may be mediated through glutamate release by DA neurons. In addition to glutamate, GABA might also be present in DA neurons and act as a cotransmitter. Indeed, immunocytochemical data shows that a subset of DA neurons in the substantia nigra compacta contains GAD (78). Indirect evidence also suggested the presence of GAD in 5-HT neurons (79). These findings are controversial considering an earlier report showed that DA neurons lack immunoreactivity for GABA in the rat (47). In primary culture, however, a small subset of DA neurons do indeed appear to contain GABA (47, 80), although they appear not to release it synaptically. We can therefore conclude that although a minority of DA (and perhaps 5-HT) neurons may express GAD and synthesize GABA, in some cases, this transmitter does not appear to act as a bona fide cotransmitter in these neurons.
Schematic representation of the synaptic and non-synaptic axon terminals established by dopamine neurons. DA neurons are known to establish two morphologically distinguishable axon terminals: some that are non-synaptic and others that are synaptic. The non-synaptic terminals (varicose-like structures) display no obvious pre- and postsynaptic specialization (see lower illustration showing a magnified view of a single non-synaptic terminal), contain tyrosine hydroxylase (TH) and could be specialized for the release of DA. The synaptic terminals display a more classical active zone, postsynaptic density and synaptic cleft, and could be the site of VGluT2 expression and of glutamate (Glu) release (see upper illustration showing a magnified view of a synaptic axon terminal).
Can Neurotransmitter Phenotypes Change in Response to Changes in Activity or Disease?
Following initial work on the role of activity and calcium influx on neurotransmitter phenotype choice in sympathetic neurons (81), an increasing amount of evidence suggests that during both development (in utero) and the postnatal period, the neurotransmitter phenotype of neurons is plastic and can be adapted as a function of activity or various environmental signals. Thus, activity-dependent phenotypic plasticity occurs in mature animals, suggesting that a dormant phenotype can be put in play by external inputs. A few examples are briefly reviewed here.
Activity-dependent Homeostatic Changes in Neurotransmitter Phenotype
Neurons of the Xenopus laevis embryonic spinal chord gradually give rise to the cholinergic, GABAergic, glycinergic, and glutamatergic neurons found in adult animals. The neurotransmitter phenotype of these neurons is influenced by their spontaneous firing activity and by the intracellular calcium spikes associated with firing (82, 83). Silencing of these developing spinal neurons by overexpression of potassium channels or by blocking calcium spikes with sodium and calcium channel antagonists enhances the expression of cholinergic and glutamatergic markers (choline acetyltransferase and VGluT2, respectively) whereas enhancement of their activity by overexpression of voltage-dependent sodium channels or by using the sodium channel activator veratridine inhibits the expression of these same phenotypic markers. The neurotransmitter phenotype plasticity observed in this model appears to be homeostatic in terms of maintaining an adequate balance of excitatory and inhibitory transmission because the increased or decreased expression of the cholinergic and glutamatergic phenotypes was accompanied by opposite changes in the proportion of neurons containing GABA or glycine. Motoneurons normally expressing cholinergic markers can thus begin expressing GABA in response to increases in calcium spiking. Interestingly, plasticity of neurotransmitter phenotype in these neurons is accompanied by corresponding adaptations in the post-synaptic receptor repertoire (84).
Transmitter Phenotype Changes in Hypothalamic Neurons
Basal co-release of glutamate and GABA from a set of neurons is documented in the hypothalamus of adult female rats during the ovulation cycle (85). Indeed, cells in the anteroventral periventricular nucleus of the hypothalamus, which are mainly GABAergic, express glutamatergic markers. Furthermore, the expression of markers of both phenotypes is regulated by photoperiodic signals and estradiol stimulation. In addition, phenotypic plasticity of a type similar to that found in spinal neurons (discussed above) has also been described in hypothalamic neurons. When placed in primary culture, embryonic neurons of the medial hypothalamus display a range of neurotransmitter phenotypes, including glutamatergic, GABAergic, and dopaminergic. A small proportion (less than 1%) of cholinergic neurons is also found. Interestingly, chronic blockade of NMDA-subtype glutamate receptors or calcium-channel antagonists induces a marked increase in the proportion of neurons expressing choline acetyltransferase, associated with a large increase in ACh-induced excitation and calcium influx in neurons (86, 87). Although the physiological significance of this observation has not been closely examined [but see (88)], the data show quite clearly that a subset of hypothalamic neurons have the potential to modify their neurotransmitter phenotype in response to chronic changes in the release or synaptic action of classical neurotransmitters.
Magnocellular vasopressin and oxytocin-containing hypothalamic neurons also demonstrate neurotransmitter phenotype plasticity. Increasing the concentration of sodium chloride in the drinking water of rats induces an increase in the expression of VGluT2 mRNA and protein in these neurons (89–91). These data suggest that glutamate co-release in this neuronal population is highly plastic even at postnatal stages in response to physiological stimuli.
Glutamate-GABA Co-release in Epilepsy
Epileptic activity increases the GABAergic markers in the glutamatergic granule cells of the hippocampus: expression of GABA, GAD67, and vesicular GABA transporter (VGAT) mRNAs is elevated after seizures (28, 92–94) (Figure 1⇑). and under these conditions, activation of the mossy fibers in the presence of ionotropic glutamate receptor antagonists produces GABAAR-mediated monosynaptic responses (29, 30, 95, 96) and the release of GABA from the mossy fibers produces a tonic GABAAR-mediated inhibition of spontaneously occurring oscillatory activity in CA3 (97). Interestingly, the presynaptic modulation of glutamate and GABA release from the mossy fibers appears to be controlled by different metabotropic glutamate receptors (33, 98). Epileptic activity is generated when the balance of excitation and inhibition is impaired and when excitatory activity—normally conveyed by cells releasing glutamate—surpasses inhibition (normally exerted by cells releasing the inhibitory amino acid GABA). Thus, because a tight maintenance of the excitation-inhibition balance is needed to avoid the emergence of epileptic activity, the emergence of GABAergic activity from an originally glutamatergic pathway would provide an effective means to rapidly auto-regulate the system by limiting excitability (30, 97). After the seizure-evoked emergence of the GABAergic phenotype in granule cells—and thus, in the mossy fiber pathway—the spontaneous oscillatory activity of area CA3 is modulated in a manner consistent with tonic GABAA-receptor-mediated inhibition, whereby β/γ oscillatory activity is depressed, probably to limit further the spread of hyperexcitability (97). Oscillatory activity in the β/γ band (20–80 Hz) is thought to be of key importance for higher brain functions. Oscillations provide a temporal framework, with regards to which neurons may either fire in synchrony or not. The synchrony of firing carries meaning, in that such synchrony (or its absence) defines whether particular groups of neurons are devoted to a common task. Thus, inhibition of oscillatory activity in CA3 by enhanced GABA transmission from the dentate gyrus could, in part, underlie the deficits in storage of information in the hippocampal network after seizures.
Are There Advantages to Cotransmission?
Functional cotransmission resulting in the release of two or more neurotransmitters by a single neuron implies that postsynaptic receptors for both transmitters exist in front of or in the vicinity of the terminal. In the context of such a configuration, a neurotransmitter could eventually modulate the action of another at either side of the synapse. The existence of different postsynaptic receptors apposing a single terminal could allow differential control of the excitability of a given neuron and the receptors for each neurotransmitter could also be differentially modulated. It is possible that co-release adds a safety factor to the communication process. Such a mechanism could compensate for activity- or pathology-dependent alterations in postsynaptic receptor subunits. Whether and how this actually occurs in various neuronal populations is an open field of research.
Current Challenges
The work reviewed here illustrates the fact that the neurotransmitter repertoire of neurons is highly plastic, and it is likely that in the near future, a number of additional examples of neurons co-releasing classical neurotransmitters will be described. One of the major challenges that lie ahead will be to identify the physiological and pathophysiological signals that regulate such plasticity in adult animals and in humans.
- © American Society for Pharmacology and Experimental Theraputics 2007
References

Rafael Gutiérrez, PhD, is a Professor of Neurosciences at the Center for Research and Advanced Studies in Mexico City. He described the emergence of GABAergic transmission from the glutamatergic mossy fiber pathway after the induction of hyperexcitability in the adult dentate gyrus and the transient expression of the dual glutamatergic-GABAergic phenotype in the developing granule cells during development. His research interests include the molecular determinants of phenotypic plasticity of the granule cells and its consequences in hippocampal function. E-mail gra-fael{at}fisio.cinvestav.mx; fax (5255) 5061-3368.

Louis-Eric Trudeau, PhD, is an Associate professor of pharmacology, neuroscience and psychiatry at the Université de Montréal Faculty of Medicine in Montreal, Quebec. His research interests focus on the regulation of neurotransmitter release by dopamine neurons of the midbrain. His group has been recently studying some of the peculiar properties of these neurons, such as their ability to release dopamine from their somatodendritic compartment or their capacity to use glutamate as a cotransmitter at some of the axon terminals. E-mail louis-eric.trudeau{at}umontreal.ca; fax 514-343-2291.