Anti-Oncomir Suppression of Tumor Phenotypes
Tumors arise through the accumulation of genetic mutations and epigenetic alterations that result in the inactivation of tumor suppressor genes and the increased expression of proto-oncogenes. Only recently has it been appreciated that expression of microRNAs (miRNAs)––small (~22 nucleotide) non-coding RNAs––is frequently altered in tumor tissues (1–4). miRNAs can have pleiotropic effects on cell proliferation, apoptosis, and cell differentiation. They are generally believed to act by binding to imperfectly complementary sequences in the 3′ untranslated region of target genes, resulting in decreased translation or in the degradation of the target transcript (5, 6). Bioinformatic and experimental evidence indicate that each miRNA may target several dozen to as many as several hundred gene transcripts, thus allowing for coordinated and combinatorial regulation of multiple genes by miRNAs (7).
A growing body of evidence implicates miRNAs as regulators of tumor phenotypes (8–12). Perhaps the strongest link for this has perhaps been made by Golub and colleagues (2), who showed that expression profiling of ~200 miRNAs provides a more accurate approach to distinguish the tissue of origin among poorly differentiated human cancer types than does the expression profiling of >20,000 mRNAs. The close correlation between the miRNA expression signatures of a relatively small number of miRNAs and their corresponding tumor phenotypes suggests that particular miRNAs may play a causal role in tumor formation and maintenance.
miRNAs are deregulated in a variety of human cancers; however, their precise role in cancer remains largely obscure. On the one hand, global loss of miRNA expression by the targeted deletion of Dicer, an miRNA-processing enzyme in mice, appears to permit, indeed even to stimulate, tumor formation in a K-Ras-driven lung tumor model (13). In contrast, loss of the Dicer gene or of a gene encoding a different miRNA-processing enzyme [DiGeorge syndrome chromosomal region 8 (DGCR8)] from the Drosophila melanogaster germ line also results in the loss of global miRNA production; however, the loss of either of these two is associated with increased accumulation of germ cells or embry-phase of the cell cycle, reducing the onic stem cells in the G1 proliferation rate of these cells (14). Thus, global loss of miRNAs can either slow proliferation or cooperatively promote tumor formation, depending on the cellular context (i.e., cell type–specific effect) or the cancer model system tested.
There are a growing number of examples of particular miRNAs whose expression is increased in cancer (i.e., mir-21, the mir-17-92b cluster, mir-155, and mir-372/373) whereas the expression of many other miRNAs is decreased in tumor versus normal control cells. miRNAs that have been experimentally shown to directly induce tumor phenotypes (i.e., formation) have been termed “oncomirs” (8), with the first example representing the mir-17-92b cluster. This coordinately expressed cluster of miRNAs can cooperate with the MYC oncogene to increase the incidence of lymphomas or increase tumor angiogenesis in experimental mouse model systems (8, 15).
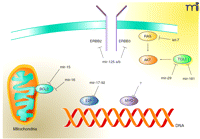
In contrast, particular miRNAs may function as tumor suppressors by targeting oncogenes, thus attenuating their expression and antagonizing tumor formation. We refer here to this class of miRNAs as “anti-oncomirs.” Examples of oncogene–anti-oncomir pairs include: RAS–let-7a (10), BCL2–mir-15 and BCL2–mir-16 (16), and ERBB2/ERBB3–mir-125a/b (17) (Figure 1⇓). Regulation of RAS expression by let-7a is noteworthy because this oncogene–miRNA pairing has been conserved from C. elegans to humans, implying that oncogene–anti-oncomir pairs have co-evolved to regulate developmental programs. Also noteworthy, the related miRNAs mir-125a and mir-125b can coordinately regulate ERBB2 and ERBB3, representing the first example of a single miRNA targeting two molecules in the same signaling cascade (17). The mir-17-92b cluster functions as both an oncomir and anti-oncomir: in addition to its ability to cooperate with MYC, mir-17-92b can negatively regulate the expression of E2F, a transcription factor required for cell-cycle progression (11). Finally, although no specific miRNA has yet been identified that directly targets MYC oncogene expression, MYC is likely a target of regulation by anti-oncomirs because loss of Dicer enzyme results in increased MYC expression (13).
Another oncogene–anti-oncomir pair was recently added to the list by Croce and colleagues, who showed that the TCL1 oncogene is targeted by mir-29 and mir-181 (18). TCL1 is an oncogene that functions as a coactivator of the serine–threonine kinase Akt and is overexpressed in a subset of T-cell leukemia/lymphomas and chronic lymphocytic leukemias (19). Elevated TCL1 expression is frequently observed in an aggressive and chemotherapy-refractory form of CLL that is associated with deletion of a portion of chromosome 11q (20, 21). Profiles of miRNA expression from CLL patient samples identified a number of miRNAs whose expression is decreased in the aggressive CLL variants that harbor the 11q deletion. Using both luciferase-based reporter assays and Tcl1 protein expression analysis, the authors demonstrated decreased expression of Tcl1 by forced expression of either mir-29 or mir-181 (18). Interestingly, several groups have recently demonstrated that expression of mir-29 is induced by p53 expression, providing further support for its role as anti-oncomir. Croce and colleagues find an inverse correlation between Tcl1 protein expression and mir-29 and mir-181 expression in CLL patient samples. This study focuses attention on mir-29 and mir-181 as important regulators of TCL1 in CLL. Clearly, further work is required to determine if suppression of mir-29 and mir-181 expression is an early or late event in the genesis of aggressive CLL. Based on the model presented, it would be predicted that miRNA inhibitors specific for mir-29 or mir-181 can increase Tcl1 expression in indolent CLL cells, resulting in increased resistance to chemotherapeutics.
Although anti-oncomirs have been identified by a variety of experimental approaches, it remains to be determined whether forced expression of particular anti-oncomirs can cause tumor regression in model systems. The advent of liposomal-based or cholesterol-modified delivery systems for nucleic acids into target tissues (22) has raised the possibility of using anti-oncomirs as “natural” inhibitors of oncogenes. It is currently unclear whether the delivery of synthetic miRNA mimetics offer an improvement versus small inhibitory (si)RNAs designed to be perfectly complementary to an mRNA of interest, resulting in the cleavage of the mRNA. Because miRNAs may primarily affect translation rather than mRNA cleavage, assessing the anti-oncomir therapy will require analysis of the protein expression of an oncogene that is being targeted. Additionally, little is currently known about pharmacologic properties of siRNA inhibition in whole animals. It may be that miRNAs, by acting principally through translational regulation, offer some advantages over siRNA-based therapies. For example, there are many circumstances where a single miRNA decreases the expression of multiple different genes involved in the same pathway. Thus, in principle, forced overexpression of anti-oncomirs would have the advantage over a single siRNA by possibly targeting numerous oncogenes.
A second possible approach to exploit anti-oncomirs as tumor-suppressors is through the pharmacologic regulation of pathways which in turn can increase anti-oncomir expression. For example, we have shown that histone deacetylase (HDAC) inhibitors can rapidly increase the expression of a subset of miRNAs (23). Some of the induced miRNAs include the anti-oncomirs mir-125a/b, regulators of ERBB2. Treatment with HDAC inhibitors rapidly decreases ERBB2 expression, likely through anti-oncomirs as well as other mechanisms. Thus, anti-oncomirs represent natural inhibitors of oncogenes. Taking advantage of these pathways, however, will require the development of novel pharmacologic agents and assays to assess their effectiveness.
Targeting oncogenes by anti-oncomirs. Diverse oncogenic signaling pathways that may include anti-apoptotic measures and other signals from tyrosine kinases to transcription factors are regulated by specific miRNAs. Oncogenes and the miRNAs that target them for translational repression are indicated.
Acknowledgments
This work was supported in part by National Institutes of Health Grants P50-CA58207, K08-CA104032 (to AG) and R01-CA36773 (to CB). We thank Drs. Chris Sullivan and Noelle L’Etoile for helpful discussions.
- © American Society for Pharmacology and Experimental Theraputics 2007
References

Christopher C. Benz, MD, is the Director of the Buck Institute Program on Cancer and Developmental Therapeutics. He is an Adjunct Professor of Medicine, UCSF Division of Hematology-Oncology and Comprehensive Cancer Center. Dr. Benz earned his B.S. in biochemistry at University of California, Los Angeles (UCLA) in 1968, and M.D. at University of Michigan School of Medicine (Ann Arbor) in 1972. He completed his Internal Medicine and Oncology specialty training at the Vancouver General Hospital, University of British Columbia in 1978, and then finished his postdoctoral training and joined the medical faculty at Yale University School of Medicine. In 1983, he was recruited to join the Division of Hematology-Oncology (Department of Medicine) and Cancer Research Institute of the University of California, San Francisco (UCSF). In 2000, Professor Benz relocated his breast cancer research program from UCSF to the newly opened Buck Institute for Age Research (Novato, CA). He continues to maintain his professorship at UCSF, cares for patients at the UCSF/Mt. Zion Breast Care Clinic, and plays an active role in the UCSF Comprehensive Cancer Center’s Breast Oncology Program. As Director of the Buck Institute’s Program on Cancer and Developmental Therapeutics, Dr. Benz’s translational bench research focuses on identifying molecular strategies to improve breast cancer diagnostics and therapeutics. E-mail cbenz{at}buckinstitute.org

Andrei Goga, MD, PhD, is an Assistant Professor in the UCSF Department of Medicine, Division of Hematology/Oncology. Dr. Goga completed his doctorate in molecular biology and medical degree at UCLA in 1997. During his doctoral work he studied the mechanism of BCR/ABL signaling in human leukemia in the laboratory of Dr. Owen Witte. He completed Internal Medicine and Medical Oncology clinical training at UCSF in 2003. He is an Attending Physician at the UCSF Carol Franc Buck Breast Care Center. Dr. Goga completed joint post-doctoral training with Drs. David Morgan and J. Michael Bishop studying cell cycle regulation in cancer. In 2006, he was appointed an Assistant Professor of Medicine at the University of California, San Francisco. He is a member of the Biomedical Sciences Graduate Program, the Bay Area Breast Oncology Program and an active member of the UCSF Molecular Medicine Program. His laboratory studies oncogene and miRNA signaling pathways in cancer. Using functional screens, his research group hopes to identify novel synthetic-lethal combinations of miRNAs or oncogenes and cell cycle inhibitors. E-mail andrei.goga{at}ucsf.edu; On the Web: oncogenes.net; fax 415-476-4190.