40 Years as a Chemist at the NIH

John Daly describes his career as “four decades as a chemist at a biomedical institute.” The term certainly fits: Daly speaks fluent organic chemistry, recounting reaction mechanisms that he has been involved with, among them landmark findings such as the “NIH shift” (first observed in the enzymatic hydroxylation of phenylalanine) and the function of forskolin as an activator of adenylyl cyclase (now widely used as a tool in signal transduction research). He is a staunch proponent of the power of chemistry to answer biological questions, and privately, he expresses optimism that the biomedical community is now realizing the value of basic chemistry to the future of the pharmacological sciences. Daly’s own career, including well over 500 published papers, is brilliant testimony to the importance of chemistry in forging research paths that lead, often unforeseeably, to a broader appreciation for biological concepts, as well as to concrete tools for the analysis and treatment of human disease. He looks back at his four decades at the NIH with pride and affection for the great scientists he has worked with, and he argues for the importance of the NIH as a continuing model of intellectual collaboration across biomedical disciplines.
MI: So, you see yourself primarily as a “chemist?”
JD: Well, I got into the National Academy not as a chemist, but as a pharmacologist. Because as a chemist, I’ve tried to go beyond just coming up with compounds; I’ve also tried to identify biological targets—sometimes to identify valuable research probes.
MI: Did you get into science then, planning on a career in pharmacology?
JD: My undergraduate degree was in biochemistry. Back then, the term “pharmacologist” wasn’t bandied about as much as it is now. In the school I went to (Oregon State), biochemistry was the discipline closest to understanding the functions of cells and organisms. What turned me on to organic chemistry was a very good organic chemistry professor, but I recognized in the biochemistry courses that I took that the people had little idea of the relationships between chemical structure and biological function. They would have to look at the board and laboriously draw the structure of something like ATP. But to me every aspect of chemical structure was interesting for its potential to interact with biological entities. One of the courses that really intrigued me was embryology, because it had then become clear that the notochord causes differentiation in surrounding tissues; there was obviously communication between cells, and I found that very intriguing. So for my senior project, I was making purines for testing as anticancer agents, but I wanted to see if they worked in embryonic cell signaling.
MI: What did you know about purines in signaling at that time?
JD: Actually, nothing. So little was known! That was back in the mid 50s. My organic chemistry professor, Bert Christensen, talked me into applying for an NSF fellowship and doing a master’s with him. I wrote up a proposal that I was going to make compounds and then I was going to find an embryologist to collaborate with and see if these compounds would affect differentiation. I’m sure the committee looked at my proposal and said, “This kid doesn’t know what he’s talking about, but at least he’s thinking.” And we’re still trying to decipher signaling and the actions of growth factors that control gene transcription.
MI: So you were interested in questions of how information flowed in biological systems.
JD: Yes, but I never got an embryologist to work on this with me, although I did find that some compounds had some anticancer activity. I ended up getting a master’s in pure organic chemistry.
MI: And you did that in only a year—that was also at Oregon State?
JD: Yes. But then I found myself in a dilemma. When I applied to pharmacology departments for my doctoral work, I didn’t get accepted. Because they thought, “This kid doesn’t know what he wants to do—he did biochemistry, then organic chemistry, and now he wants to do pharmacology?”
MI: Do you think your rejections reflected how pharmacologists were defining pharmacology at that time?
JD: No, I just think I was perceived as somebody who couldn’t make up his mind. So I applied to three chemistry departments on the west coast and got accepted to all of them, and I decided to go to Stanford. My major professor at Stanford was Richard Eastman, who was a good mentor to me. His approach was to offer incoming students a number of projects to choose from. The project that appealed to me most had to do with an unknown compound from peppermint oil. I was to isolate it and determine its structure.
MI: Was this compound viewed as a potential odorant or flavoring?
JD: No, it was just a very interesting chemical in terms of its reactivity, which was the real reason for studying it. It was very labile. This was back before NMR was really established. So, I had to construct my own gas chromatograph and build my own Wheatstone bridge detector. I started identifying the degradation products and comparing them to a library of known terpenes. We finally came up with the structure and reaction mechanism, but it was a project that would probably now take only a few days. It was kind of the last days of doing classic degradation chemistry, which had always been the major strategy of chemists.
MI: So you saw yourself in graduate school as a classical chemist?
JD: Well, maybe I also picked that project because it had to do with a natural product. And having finished my PhD in chemistry, I knew I wanted to go somewhere to really get into pharmacology.
MI: And what did that mean to you, to “get into pharmacology” then?
JD: Well, to start looking at compounds in various systems, with enzymes—I don’t think the term “receptor” was really used then.
MI: So pharmacology to you as a chemist meant enzymology?
JD: To a degree, but it also meant looking at how compounds would affect cells. I think I’ve always been interested in looking at whole cells.
My professor (Eastman) knew of my interests and encouraged me to approach Bernhard Witkop here at the NIH. Witkop gave me a postdoctoral position, and he was very good about giving you complete freedom in your projects. He also allowed me to go out and start collaborations independent of him. A couple of my projects resulted in my working with Julius Axelrod, and that was really pivotal in my career. Working with Julius, I got into doing assays with catechol-O-methyltransferase. At that time, people weren’t really coming at pharmacology from a chemist’s viewpoint. What was being done required electrophysiology and organ bath preparations. And I certainly didn’t have the time or the background to set that up. But with Julius, I got so much guidance in terms of setting up an experiment that was simple and yet would yield an answer.
So we did a lot on methylation pathways over my first three years here. The other thing that was nice about the old days of NIH was that people that came here as postdocs could often look forward to having a career here. That doesn’t really occur any more. We’ve changed to more a university-type atmosphere, where new permanent positions are advertised as tenure-track positions.
When I came here, there were many people who came to NIH and did their whole career here, starting as postdocs. And even then it had sort of an aura about it as a great place to be. I came in with a two-year appointment, and in the middle of the second year, Dr. Witkop simply said, “Would you like to be a permanent member of our group?” And I loved what I was doing here, so I said I would. But nowadays, if you were to come on to NIH staff that way, you’d have to go before a committee and there would be a lot of correspondence and recommendations written. In my early days here, a call to Ed Rall, who was scientific director, was practically enough to make someone permanent, and many of the people who became section chiefs, lab chiefs, and directors came through that way.
MI: So you really started out functioning as an organic chemist among some of the greats in pharmacology.
JD: Yes, I still consider myself an organic chemist—that is the viewpoint that I bring to pharmacology. I became very interested in the whole field of biogenic amines, which got me interested in what was soon to be called receptors for these compounds. And it got me hooked into asking, “Is there some simple way that I can look, without organ baths and so on, at these interactions?” So with Bob Creveling who had been in Sydney Udenfriend’s group, we developed a mouse assay system where you gave the mouse radioactive norepinephrine and it labeled the heart, and then you’d inject various compounds and see if they’d cause its release, and you had a very nice assay where each mouse was a point. And you could do dose-response curves. And then I also worked with Creveling on some of the metabolic enzymes, looking, again as an organic chemist, for inhibitors. So, I was able to follow a whole variety of interests.
MI: All of which together add up to “pharmacology”?
JD: Yes, and then another turning point in my career had to do with the use of frogs in Colombia, providing poison for blow darts.
MI: Pharmacology led you to Colombia—to study frogs?
JD: It goes back to Witkop. He had trained in Wieland’s lab in various curare alkaloids used to poison arrows, and toxic substances from Amanita mushrooms. The story that I remember is that he was contacted at some sort of cocktail party by Marte Latham, who pointed out that Colombian Indians were poisoning their darts with frogs. Witkop found this very exciting, and so he sent a postdoc, Fritz Märki, down there to work with this woman and to get some frogs. And so they got the extracts and discovered that there was an extremely toxic substance that looked like an alkaloid; it turns out that there were really three toxic alkaloids. Witkop and Märki published a brief paper, and I got involved then.
MI: How were alkaloids defined then?
JD: Alkaloids were something at that time known exclusively from plants. So, to get them from the skin secretions of a frog was incredible. And it’s kind of strange because they didn’t make a big deal of that in the papers.
MI: And then you worked out the toxin structures?
JD: Well, they didn’t have enough material from the first expedition, and Fritz was going back to Switzerland, and Witkop approached me about the project and said, “Looks like we have a very exciting toxic substance here. I hear it’s very dangerous to go down into the rain forests of Colombia.” I’ve always been interested in the outdoors and in nature in general, so it sounded like a great opportunity to me.
MI: And the adventure—was that something that appealed to you?
JD: Well, no, it was that the project sounded very interesting (and it really was not as dangerous as we had been led to believe). Here was a very toxic natural product—I wanted to know how it worked! That was kind of different from how a lot of natural product chemists were thinking—they were looking for unique structures, without caring that much about biological targets. But I’ve always been interested in how compounds cause physiological effects. So off I went to Colombia with this Marte Latham. She had many projects. After we had gone down, she interested National Geographic in doing a photo of natives and skinning frogs. She also had a project where she was collecting these giant earthworms as a source of collagen for another group here at the NIH.
MI: Did you witness the native use of the frog poison for hunting?
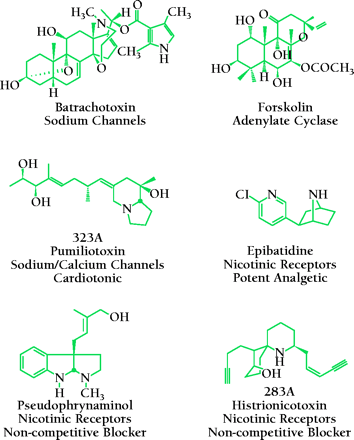
JD: Yes, but the Indians weren’t very good collectors. They weren’t that interested in the money we were paying for frogs. But we were staying in a village called Playa de Oro, and villagers there were our main collectors of the frog (Phyllobates aurotaenia). I hate to get into the number of frogs I killed in my life. The toxin was very stable, but the three major compounds (batrachotoxin, homobatrachotoxin, and batrachotoxinin-A) were collectively present at only 300 micrograms per frog, and back in those days you needed about 10 milligrams of material for analysis. Anyway, we got the mass spec data for the three compounds and tried to put structures together. Well, we were misled by the mass specs. There were no computers then—you matched peaks manually to calculate exact masses. Finally, Takashi Tokyuama, my first postdoc, obtained crystals for x-ray analysis, and Isabella Karle gave us the x-ray structure of the steroid part. Tokyuama made all the methyl esters of the pyrrole part of the batrachotoxin and nailed down the structure by NMR. Then when he synthesized batrachotoxin, he came running into the lab and—sneezing violently—says to me, “John, I think I’ve done it!” It was extremely toxic, but became very useful all over the world to probe how local anesthetics, anticonvulsants, and antiarrhythmics interact with the sodium channel that Edson Albuquerque showed to be the target of the toxin.
MI: Eventually you described over 500 alkaloids. What other organisms did you begin to look at?
JD: Well, on that first trip, I wandered off to look around the village, and I discovered another frog there that I knew nothing about. It was also brightly colored, and I made extracts from it, which turned out to contain very different alkaloids. But the problem with following up on this second species (which turned out to be Dendrobates histrionicus) was that I didn’t know where to go for large quantities of frogs. But then a lucky thing happened: Medical World News got interested in our story of the batrachotoxin structure and its potency on sodium channels—and so, they published a blurb about our work. Charles Myers, a grad student at that time down in Panama, was doing his thesis in herpetology and saw the news release. He wrote me and said there were several populations of frog named Dendrobates pumilio in Panama that were also brightly colored. [The frogs shown here illustrate the various populations of color morphs of the species.] He spoke about the evolutionary implications of frogs advertising their own toxicity—that they evolve a toxin as well as a color to advertise toxicity. He said that he expected that the more brightly colored frogs would have more toxin, and so I began collaborating with him. On my next trip to Colombia, I stopped off to see Myers, and we collected several populations of these brightly colored frogs. I got back and discovered that a particular brightly colored frog was highly toxic, but that some of the brightly colored frogs were hardly toxic at all. We now know that those frogs don’t produce the toxins at all; rather, they acquire the alkaloids through their diet and store them in skin glands. Frogs that don’t feed on the appropriate toxic arthropods fail to become very toxic. Anyway, Myers and I became close friends, and he became my mentor in herpetology.
MI: And you found that the different species—those in Panama as opposed to those in Colombia—contain different toxic alkaloids?
JD: Yes, even in Colombia we got different compounds from different frogs: we found that the D. histrionicus contained a compound distinct from the batrachotoxins—the difference in location accounting for the difference in the toxin that was accumulated within the frog. Albuquerque determined that the histrionicotoxin blocks nicotinic receptors, and histrionicotoxin has become kind of the classical high-affinity blocker for those receptors. And Myers and I started looking at other frogs and so we’ve found many compounds unusual in biological systems. And now I’m invited to herpetology meetings as a keynote speaker.
MI: And do you enjoy the herpetology now—is that a scientific interest?
JD: Oh yes, I enjoy the fieldwork. And when I go with Myers, he as a practicing herpetologist has to do a lot of photography, and together the two of us have described about nine new species of frogs. In fact, through a Polish anthropologist, we were told that there was another frog in Colombia that was being used to poison blow darts and they didn’t even need to kill the frog to apply the skin secretions—they would trap it with a stick and just scrape several blow darts across its back. And each of those was sufficient to kill a person. So, we named that the “terrible frog,” Phyllobates terribilis, because it had ten times as much toxin in it as the other two poison dart frogs. I’ve been supplying researchers around the world for the last thirty years from a single stock.
So anyway we’ve now detected and defined the structures of hundreds of compounds, and we’ve found about thirty arthropods, on which the frogs feed, that contain the compounds. We’ve looked at over ninety species of frog. We hope to find all of the compounds in arthropods—its just that the frogs are better bioprospectors than we are. Without the frogs accumulating compounds, the toxins would not have been discovered.
MI: Have any of these compounds led directly to pharmaceuticals?
JD: Well, there’s been some use of the batrachotoxins in spinal anesthesiology. And certainly the compounds we have described have been used as probes by drug companies to understand drug action on the sodium channels. For the pumiliotoxins, I think there’s real potential there as cardiotonics—as a stimulator in congestive heart failure in cases where digitalis is now used. If you modify some of the chemical functions on these toxins, you can get compounds that are cardiodepressants. There were a couple of European studies of the histrionicotoxins in kidney treatment. A drug that has been developed is based on the analgesic epibatidine. Analogs of it are still in development and are very exciting.
MI: And epibatidine came from where?
JD: From one of our exploratory expeditions down to Ecuador in the late 70s. An Ecuadorian frog, D. histrionicus, had a unique histrionicotoxin that targeted both nicotinic channels and sodium channels, and Albuquerue was interested in it, and so we went down to get large quantities. Myers and I also collected another frog, named Epipedobates tricolor. I came back with some extract of that frog and, as I usually do, I injected it into a mouse. But this time, I said, “Wow, that’s the Straub tail!” I knew from pharmacology that opioids cause this characteristic Straub tail—an arching of the animal’s tail over its back. The Straub response used to be a test to detect doped racing horses. So I was really excited, because none of our other extracts did this. We made a few more expeditions, which were not that successful in terms of finding more epibatidine-containing frogs. We did not have enough of the active compound, which we named epibatidine, to determine the structure. Then, in 1990, my long-time colleagues Martin Garraffo and Tom Spande and I started brainstorming as to how to use our limited supply of epibatidine and determine its structure by the greatly improved NMR techniques. Martin Garraffo came up with a neat way of isolating the alkaloid and we got the structure of this potent analgesic and published it in 1992. In 1993, Nobel laureate E.J. Corey called me up it to say that his group had synthesized epibatidine and some related compounds. And Corey was great—he asked if he could send us the compounds so that we could start looking at their pharmacology. And so we found that epibatidine interacts with several nicotinic receptors, so that we demonstrated that it was a full agonist—the most potent known—at those receptors.
Well, to sum up, we’re still examining alkaloids. We’re looking very hard at arthropod sources. We call the field “combinatorial bioprospecting,” where we have to look at environmental sources, including microbial species, that may be required together to produce a given alkaloid. And we’re trying to trap insects. We did find some ant alkaloids. But because the frog keeps these alkaloids in the skin for years, it would be hard to pinpoint a dietary source that would be available to the frogs only during limited times or places.
MI: It sounds like a lot to ask of an organic chemist!
JD: Well, it’s been a lot of fun. Unfortunately, it became impossible to return to Colombia for frogs due to Colombian regulations. But then back in about 1990, I got a letter from a grad student, Jack Dumbacher, who was working in Papua New Guinea. He was convinced that he had found a Pitohui bird that was toxic: When he handled it, it caused numbness, and after handling feathers, he put his finger to his mouth and it really burned his tongue. He said that the natives think it’s poisonous, although if you’re really hungry, you can skin and then rub the bird with charcoal, wash the charcoal off, and cook it (laughs). I was kind of dubious, but I told him to get some feathers and skin and put that into methanol, and then get some other tissues. I got my mouse assay going and wow, it was toxic: The mouse went into convulsion and died. I immediately did a fractionation and it turned out to be an alkaloid! So I was really excited, but, despite its potency, there was very little of this substance. I wasn’t thinking of batrachotoxin: Why would a new-world toxin suddenly turn up in birds half way around the world where there weren’t any toxic frogs? But on the mass spec it had the molecular weight of 400, and the spectrum clearly showed that it was homobatrachotoxin. We’ve been trying for about six years to bring the birds here but haven’t yet. The birds always die, once they’re caught, because of the time necessary to get the regulatory paperwork finished. [Another batrachotoxin-containing bird, Ifrita kowaldi, is illustrated at the beginning of this article.]
MI: So, from being a chemist, your work in pharmacology has brought you into herpetology as well as ornithology!
JD: Well, yeah. You bring a different approach than somebody who’s not trained in chemistry. This is a point in history where the interaction between chemistry and biology is so ripe for the picking. There are wonderful new approaches in chemistry. The Nobel laureate from just last year, Barry Sharpless, has come up with something that they call click chemistry, and this will be very powerful for probing biological systems. To me, it is just one of many incredible techniques that everyone in the world is going to start picking up. It’s one of the areas where chemical biology can really prosper. And you really need a chemist as part of the team. Our institution (NIDDK) is launching such a chemical biology initiative and I and my collagues look forward to being involved in it for the rest of our scientific careers.

- © American Society for Pharmacology and Experimental Theraputics 2002



