Roadmap or Roadkill: A Pharmacologist’s Analysis of the NIH Molecular Libraries Initiative
There is in all of us a psychological tendency to resist new ideas.
—W.I.B. Beveridge, The Art of Scientific Investigation
Introduction
Sometimes it is difficult to remember biomedical research before the days of Web-based searches of hundreds of genes or genomes with publicly available free software. In the wake of the enormous success of the genome sequencing projects, we often forget the controversy first surrounding the funding of the Human Genome Project (1–3). In 1989, Palca reported: “This has not been a good year to apply for a grant from the National Institutes of Health.…Everywhere you turn someone has a horror story about a senior scientist who has lost funding or [about] a promising postdoc unable to get off the mark with NIH.” I was among those investigators who joined in the chorus at the time warning that the entire NIH RO1 funding program was in jeopardy.
Today, a heated public debate over proper funding mechanisms for basic biomedical science has again been precipitated by new NIH policies (4, 5). At the center of at least a portion of this debate is the NIH Roadmap, which has been labeled “extravagant” (6). Certainly, the NIH has a fiduciary responsibility to allocate moneys for the purpose for which they are requested from Congress, but public discussion and debate on allocation of NIH funds will be inevitable (and perhaps even productive). As an academic pharmacologist who has been funded primarily by the NIH RO1 mechanism for the last twenty-five years, I take a great interest in the NIH Roadmap and the impact it may have on my career, as well as the future careers of my students.
The overall purpose of the NIH Roadmap, as stated on the Web site (http://nihroadmap.nih.gov), is to provide a framework for the priorities of the NIH as a whole in optimizing its entire research portfolio. Through the Roadmap initiative, the NIH has identified three main areas of opportunity: new pathways to discovery, research teams of the future, and re-engineering the clinical research enterprise. Within the Research Teams of the Future program is a particular emphasis on high-risk research, interdisciplinary research, and public-private partnerships. These are all areas that have traditionally been core strengths of pharmacology. By the very nature of the topics addressed, pharmacologists have historically been required to engage in multidisciplinary approaches, and the tools we use come often from the private sector, so that pharmacologists have become quite accustomed to formal public-private partnerships. Re-engineering the clinical research enterprise should provide clinical pharmacologists with unique opportunities to engage in new programs and outlets.
Perhaps the most interesting component of the Roadmap initiatives is the New Pathways to Discovery program. It comprises efforts to define biological pathways and networks, develop new tools to interrogate biological systems, inventory protein structures, generate software and data management tools, and create materials and devices designed at the molecular or atomic level. Indeed, a major component of the New Pathways to Discovery is the Molecular Libraries Initiative, which is my present focus.
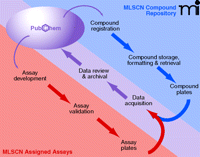
The mission of the Molecular Libraries Initiative is to empower the research community to use small-molecule compounds in their research, possibly as tools to perturb genes and pathways, as imaging probes in basic or clinical applications, or as starting points to the development of new therapeutics for human disease (http://nihroadmap.nih.gov/molecularlibraries). To achieve this goal, the NIH has established nine extramural and one intramural center as part of the Molecular Libraries Screening Centers Network (MLSCN) (7). I am fortunate to be a Principal Investigator on one of these funded programs, as are three other pharmacologists: Raymond Dingledine (Emory University), Gary Piazza (Southern Research Institute), and Dave Weaver (Vanderbilt University). The goal of the MLSCN is to conduct twenty assays per year with more than 100,000 compounds; the data generated through these assays will be deposited in the NIH-sponsored, publicly accessible database called PubChem (Figure 1⇓). PubChem currently has more than eight million compounds in its database, many of which are annotated with biological data.
Anticipated operations flow chart of the Molecular Libraries Screening Centers Network. Compounds are provided by the NIH Small Molecule Repository. Assays are assigned to individual MLSCN Centers by the MLSCN Steering Committee after peer-review. High-throughput assays are conducted and the data are rapidly deposited in PubChem. Reiterative interactions among Network components will support the development and refinement of biological activities through the synthesis of new compounds and secondary assays.
The MLSCN comprises ten diverse Centers that have various platforms and skills (Table 1⇓). This diversity permits the NIH to assign assays to each Center according to its particular expertise and technology. Each Center is required to deposit the results of their screen in PubChem, which is accessible to the public at no cost, and as of October 2006, the results from forty-five assays have been deposited. These assays range from homogenous mix-and-read enzyme assays to more complex phenotypic assays. Each Center also has the capacity to generate a limited set of analogs for further refinement of the lead chemical structure.
Core Competence of MLSCN
Roadmap Research: Grants in Neurobiology
Roadmap: The Proponent’s View
The ultimate goal of the NIH is understand and control human pathology. The proponents of this NIH experiment believe that one bottleneck is the lack of small-molecule tools for biologists to complement current genetic approaches. The MLSCN effort may afford biologists with full, partial, inverse and reverse agonists or antagonists that are reversible and active on cells or animals and that can be widely used to test contemporary hypotheses. Molecular targets that heretofore have been viewed as intractable may now be systematically addressed by the diverse collection of academic investigators in the MLSCN. Furthermore, targets associated with the more than 6,000 orphan diseases, as well as neglected diseases such as malaria and leishmaniasis, common in developing nations, are generally not attractive to commercial organizations because of the perceived potential for low revenues. The lack of a profit motive provides the MLSCN with considerable flexibility in the targets and diseases it can address.
Roadkill: The Opponent’s View
Many researchers have argued that the directed, centrally managed Roadmap research is siphoning funds away from traditional investigator-initiated competitive grants. In addition to the philosophical issues associated with funding discovery- and reagent-generating grants, there are concerns that the MLSCN may be wasting precious federal research funds (4, 5). There is also some suspicion about the willingness or ability of academic centers to deposit data rapidly into the public PubChem database. Because of the evolving nature of the MLSCN, issues associated with publication and attribution continue to pose a challenge. Some believe the data release policy might retard assay submissions to the MLSCN. Most academic centers lack the medicinal chemistry experience found in the commercial sector that is needed to refine lead structures into biologically useful substances and to scale up synthesis to enable full biological characterization. Moreover, publicly available chemical informatics to facilitate the navigation of large data sets must be developed. As with any new program, the critical path for generating and optimizing small-molecule probes has not been fully developed among the nascent Centers.
The Dangers
The viability of the MLSCN will require full participation of biologists from all fields, but there is a danger that the diverse assets provided by the Roadmap will not be adequately exploited by academic investigators. The broad academic community generally has not been exposed to the rigorous requirements for high-throughput or high-content screening, including highly automated equipment, such that many investigators may face difficulties in formulating suitable assays. Additionally, the relationship between the MLSCN and the existing or evolving academic screening centers that are outside of the Network has not been defined. In some cases, a profit motive may restrict interesting science or assay submission.
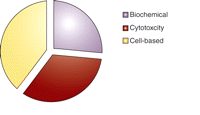
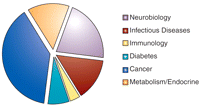
Amassing a chemical repository is a complex process, requiring judgments about compound diversity, stability, and solubility. For targets with structural information, compound libraries can provide a rapid route to structure–activity information. Although these libraries permit greater interrogation of phar-macophores, the broad variety of assays will include many instances in which there will be no known pharmacophore or target structure. The forty-five assays so far deposited, in which 801,085 compounds have been tested, are diverse (Figure 2⇓), and the twenty-eight approved NIH XO1 assays, which are provided to the Centers, span many disease categories (Figure 3⇓). The NIH and members of the MLSCN are actively reviewing the nature of the compound collection that will be supplied by the NIH. The cost of screening complex assays may restrict the total number of compounds tested. Desired analogs may not be synthetically tractable, which would limit the ability of MLSCN to generate sufficiently diverse, selective, and potent compounds.
Completed MLSCN assays. This chart lists the general categories of assays completed by MLSCN Centers.
The Opportunities
The MLSCN offers the research community, including pharmacologists, neurobiologists, cell biologists, biochemists and physiologists, an unusual asset to help them identify small-molecule probes. In the short period that the project has been operating, MLSCN members have already demonstrated their resourcefulness in producing useful compounds, such as a selective agonist for the seven-transmembrane G protein–coupled receptor GPR30 (8), sphingosine 1-phosphate type 1 receptor agonists (9), and pyruvate kinase agonists and antagonists (10). Others are anticipated soon.
Considerable care has been exerted in developing the NIH small-molecule repository, which can only be evaluated through the NIH MLSCN mechanism (http://nihroadmap.nih.gov/molecularlibraries/grants.asp). In this way, small molecules can be immediately used by the assay provider and the scientific community to probe biological processes and hypotheses. Unlike antisense or short interference RNA, these small molecules can affect a specific function of an enzyme without depleting the macromolecule; they can additionally target non-protein substances, such as lipid, sugars, ions, protein–protein and other inter-macromolecular interactions. Because the MLSCN is primarily focusing on probe molecules rather than drugs per se, areas of chemical space can be probed that are outside of the usual parameters used to build “drug-like” chemical libraries, such as the Lipinski rules.
The NIH is currently seeking unique chemical contributions from academic chemists, which will expand the utility of the repository. The XO1 funding program provides scientists access to the MLSCN at no cost to the investigator and yields a fully validated high-throughput screening protocol that can be used by the investigator for future studies or RO1 grant applications. The MLSCN offers an excellent environment for innovative assay development, and each center will assist future NIH R03, R21, and XO1 applicants. Furthermore, the chemistry cores of each center could potentially be able to refine identified lead compounds resulting in more potent and selective agents.
Conclusion
The NIH Molecular Libraries Initiative is intended to be a bold experiment that could reshape the reagents available to pharmacologists. Both young and established investigators, in both the public and private sectors, will have assess to assay development, unique compounds, automated compound analyses, annotated libraries for lead identification, and informatics tools. The initial probe compounds that are identified will likely require refinement to have the potency, selectivity, and aqueous solubility suitable for cellular and in vivo studies, but it is expected that the annotated PubChem repository will stimulate that activity. Those of us involved in the MLSCN remain enthusiastic about the uses of the NIH initiative for hypothesis testing and RO1 grant submissions, enabling assay providers and others with valuable tools. We especially wish to provide a starting point for the time-consuming and expensive process of drug development and thereby encourage others to develop compounds for investigations into orphan and neglected diseases. Public exploitation of the data in PubChem will be necessary to optimize the formulation, pharmacokinetics, metabolism and toxicology profile of probe compounds, but the accessibility of PubChem data to the research community should reduce the initial costs for researchers who address neglected diseases. Clearly, it will be important for the MLSCN members to facilitate assay implementation and to generate viable biological probes quickly—service to the research community will very definitely be the gauge by which the Network will be evaluated as a successful component of the NIH Roadmap initiative.
Acknowledgments
The author thanks Paul A. Johnston, Robb Tomko, Pallavi Bansal and Jacqui Fiske Lazo for their helpful comments.
Footnotes
-
↵1 John S. Lazo is Principal Investigator, Pittsburgh Molecular Libraries Screening Center, and Chair, NIH Molecular Library Screening Center Network Steering Committee.
- © American Society for Pharmacology and Experimental Theraputics 2006
References

John S. Lazo, PhD, is the Allegheny Foundation Professor of Pharmacology and the Director of the University of Pittsburgh Drug Discovery Institute. E-mail lazo{at}pitt.edu ; fax 412-648-9009.