 |
 |

Unattended Home Diagnosis and Treatment of Obstructive Sleep Apnea Without Polysomnography
Eugene C. Fletcher, MD;
Jacqueline Stich, RN;
Karl L. Yang, MD
Arch Fam Med. 2000;9:168-174.
ABSTRACT
 |  |
Objective To test the effectiveness of unattended home monitoring along with automatic titrating continuous positive airway pressure (auto-CPAP) as an acceptable method for diagnosing and prescribing proper CPAP pressure for treatment of patients presenting with classic symptoms of obstructive sleep apnea (OSA).
Design Nonrandomized, prospective case study of 63 patients with a presumptive diagnosis of OSA.
Setting University hospital and veterans affairs medical center ambulatory sleep disorders clinics.
Participants Fifty-eight men and 5 women were recruited for symptoms of excessive daytime sleepiness, heavy snoring, and witnessed apnea.
Intervention Subjects with 10 or more respiratory events per hour were titrated by automatic, unattended home monitoring to an optimal CPAP pressure.
Main Outcome Measures Number of subjects able to be diagnosed by unattended home monitoring, titrated to optimal CPAP pressure, accepted an auto-CPAP machine for home use, and symptoms improved.
Results Fifty-four (86%) of 63 patients completed sufficient diagnostic studies, and in 45 (83%) of these, a diagnosis of OSA was established. Nine subjects were unable to adjust to the nasal mask for an adequate diagnostic recording, and 9 had fewer than 10 respiratory events per hour. Ten subjects with OSA could not complete a titration study. Thirty-five of the subjects diagnosed with OSA accepted the auto-CPAP machine into their home, while 30 used it for therapy longer than 3 weeks. The estimated cost of performing in-home studies was less than one fourth of the estimated cost for in-laboratory polysomnographic examinations had they been performed.
Conclusion Unattended monitoring plus auto-CPAP allows cost-effective diagnosis and CPAP titration of most patients with OSA with straightforward symptoms.
INTRODUCTION
OBSTRUCTIVE sleep apnea (OSA) is recognized as a frequent cause of symptomatic daytime sleepiness in the general population.1 The importance of this disorder as a contributor to cardiovascular disease, including systemic hypertension,2 stroke,3 and coronary artery disease,4-5 is being increasingly recognized as a cause of early morbidity and mortality in patients with OSA.6-7 As many as 4% of middle-aged men and 2% of middle-aged women may have sleep apnea syndrome, which is defined as the presence of apnea during sleep with pathologic daytime sleepiness.8 Thus, diagnosing and treating OSA is becoming a major health problem in the United States from the standpoint of physician education and vigilance as well as cost.
Polysomnography (PSMGY) with electroencephalographic sleep staging, oximetry, and respiratory monitoring is a criterion standard for the diagnosis of OSA and for continuous positive airway pressure (CPAP) titration.9-10 Disadvantages are that PSMGY is labor intensive, requiring continuous technician monitoring; is not readily available in some rural areas; and long scheduling delays may occur. Intrapatient night-to-night variability may give divergent respiratory disturbance indices (RDIs), causing reclassification of the diagnosis in up to 43% of patients with lower RDIs (5-15 respiratory events per hour).11 Through new technology, unattended home monitoring now offers an alternative to PSMGY in diagnosing patients with OSA,12-14 while automatic titrating (auto)-CPAP machines may provide an alternative to in-laboratory PSMGY in the prediction of the correct CPAP pressure.15-17 Our objective was to show that in-home, unattended diagnosis and treatment of OSA with a single device is technically feasible in that measurement of airflow interruption at night is sufficiently accurate to establish this diagnosis in patients with classic symptoms.
PATIENTS, MATERIALS, AND METHODS
Inclusion criteria for this study were (1) a history of excessive daytime sleepiness, (2) heavy, sonorous snoring, (3) witnessed apnea when bed partner information was available, and (4) agreement to be monitored at home and to use and return recording devices as often as needed to establish a diagnosis and effective treatment. Exclusion criteria included complicating medical illnesses or acute decompensation requiring hospitalization, a previously established diagnosis of OSA, or suspicion by the examiner of a complicating sleep disorder, such as narcolepsy or restless legs syndrome.
PATIENTS
Sixty-three consecutive patients of 150 screened at sleep disorders clinics (Veterans Affairs Medical Center and University of Louisville Hospital, Louisville, Ky) meeting the above criteria agreed to in-home monitoring for diagnosis and auto-CPAP titration if OSA was confirmed. The subjects were a mixture of 90% public hospital patients (Veterans Affairs Medical Center, 37 and university clinic, 20) and 10% privately insured patients from the university clinic. Seven subjects were self-referred. The presence of pathologic daytime somnolence was determined from histories taken by the physician investigators (E.C.F. and K.L.Y.) (trained in sleep medicine) and confirmed and quantitated using a score on the Epworth Sleepiness Scale18 (ESS) of 11 or higher (maximum score, 24) and on the Cleveland Questionnaire19 (CQ) of 20 or higher (maximum score, 60). Follow-up questionnaires were also completed by each subject who achieved satisfactory auto-CPAP titration and used the machine for at least 3 weeks. Corroboration of the history of snoring and witnessed apnea from a spouse, family member, or bed partner was unavailable in 5 cases because the subject lived alone.
MATERIALS
A single machine was used both as an unattended monitor and auto-CPAP (Horizon; DeVilbiss, Somerset, Pa). Wearing a soft, snug-fitting CPAP nasal mask, a built-in pneumotachograph quantitatively recorded nasal airflow, apneas, hypopneas, nasal mask or mouth leaks, and snoring.20 A 3–cm H2O pressure was required to operate the pneumotachograph in the diagnostic mode and to provide ventilation to the subject. An adhesive finger oximeter recorded continuous arterial oxyhemoglobin saturation and a chest belt with a position switch indicated body position throughout the night. On study nights, all data were digitized and recorded on a dedicated computer (Surveyor; DeVilbiss) that was downloaded into a personal computer. Respiratory events of 10 seconds or longer were scored according to the default settings of the software, where apnea was defined as less than 15% of baseline flow and hypopnea as a 40% fall from baseline flow. The sum of apneas and hypopneas was automatically divided by total lapse time to arrive at an RDI per hour of recording time (RDI-machine). In addition, analog tracings were examined (unblinded) by us with respiratory events (by pneumotachograph) visually scored (RDI-visual) and nasal mask or mouth air leak time and time during the night when not connected to the machine (hereafter, off-time; eg, trips to the bathroom) to arrive at a more accurate RDI. The automated scoring software neither recognizes off-time nor subtracts air leak time from the total recording time. Thus, the removal of nonscorable periods by hand scoring considerably increased the scores for RDIs through the shortening of total recording time.
METHODS
After discussing the study and signing an informed consent, each patient was fitted with a nasal mask, instructed how to apply the machine at home, and was given a chance to adapt to mask breathing in the clinic. On average, this took 30 minutes of one-on-one instruction by the study nurse (J.S.). If the first night's recording was insufficient, uninterpretable, or had missing channels, additional nights of diagnostic monitoring were undertaken. Following a satisfactory diagnostic study night, the auto-CPAP machine was then reprogrammed for titration (if RDI-visual score 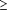 10 respiratory events per hour) with data collection and was repeated on subsequent nights until the best-effective pressure was reached (elimination of respiratory events and snoring) or the subject had substantial reduction in apneas but could not tolerate higher pressures (Figure 1). All subjects with OSA completing auto-CPAP titration were offered a multiple sleep latency test (both before and after treatment, without the previous night's PSMGY) at baseline and after a minimum of 3 weeks of therapy. 10 respiratory events per hour) with data collection and was repeated on subsequent nights until the best-effective pressure was reached (elimination of respiratory events and snoring) or the subject had substantial reduction in apneas but could not tolerate higher pressures (Figure 1). All subjects with OSA completing auto-CPAP titration were offered a multiple sleep latency test (both before and after treatment, without the previous night's PSMGY) at baseline and after a minimum of 3 weeks of therapy.
|
|

|
|
Figure 1. Left, Ten-minute segment of the analog printout from a study subject with severe sleep apnea diagnosed using the automatic titrating continuous positive airway pressure machine (Horizon; DeVilbiss, Somerset, Pa) and dedicated computer (Surveyor; DeVilbiss) as an unattended monitor for apnea and/or hypopnea. Panel 1 (top most) shows airflow from the pneumotachograph with repetitive apneas interspersed between 2 and 3 breaths; panel 2, machine scoring of apneas; panel 3, the repetitive oxyhemoglobin desaturation related to these apneas; panel 4, repeated episodes of severe snoring; panel 5, the continuous positive airway nasal mask pressure at 3 cm H20, which allows the pneumotachograph to function; and panel 6, body position. Right, The same patient studied on a subsequent night with the automatic titrating continuous positive airway pressure machine placed in the titration mode. The parameters on each panel are the same as on the left. The automatic titration upper and lower nasal mask pressure limits were set at 13 and 7 cm H2O, respectively. There was marked attenuation of apneas seen in panel 1, with normalization of arterial oxygen saturation. Panel 5 shows the nasal mask pressure being titrated upward to 13 cm H2O in response to snoring and a single apnea seen in panel 1. The subject is sleeping supine in both studies.
|
|
|
For subjects in whom a diagnosis of OSA was established by home monitoring and for whom a satisfactory auto-titrating pressure was established (OSA-TIT), auto-CPAP machines were supplied for home use (Figure 2). Those subjects whose diagnostic study results appeared adequate but showed fewer than 10 respiratory events per hour (OSA-NEG), those who had unsatisfactory diagnostic study results (UNSAT-DIAG), or those who had a diagnosis of OSA but were unable to tolerate or record satisfactory auto-CPAP titration (OSA-UNTIT) were referred and encouraged to undergo PSMGY to confirm the home diagnostic study results or to undergo titration under observation (Figure 2).
|
|

|
|
Figure 2. Schematic showing the outcome of unattended home monitoring and automatic titrating continuous positive airway pressure (auto-CPAP) machine titration in the 63 subjects. Unsatisfactory diagnosis indicates subjects whose home diagnostic study results were unsatisfactory to establish a diagnosis of obstructive sleep apnea (OSA); PSMGY, polysomnography; OSA-NEG, sufficient diagnostic study results showed fewer than 10 apneas and/or hypopneas per hour of recording time; UARS, upper airway resistance syndrome; OSA, CPAP untitrated, subjects with OSA who did not complete sufficient auto-CPAP titration studies to establish an effective CPAP treatment pressure; and OSA, CPAP titrated, subjects completing diagnostic and auto-CPAP titration studies sufficient to establish a diagnosis of OSA and effective treatment pressure.
|
|
|
The cost of home monitoring per patient was derived as follows. Technician or nurse time ($16 per hour) for nasal mask fitting and initial instruction (30 minutes) plus computer program download and reprograming time (60 minutes) plus telephone call time (10 minutes) were added to an average cost of $10 for patient travel for each study. Physician's time to read the pneumotachograph/oximeter tracing was 15 minutes ($50), all totaling $86.50 per study (diagnostic or titration). A one-time cost of $137 (retail cost) for consumables to include the CPAP nasal mask, tubing, valve, and oximeter finger piece was added per patient. Equipment costs for the 3 dedicated computer units ($4200 each) and 3 auto-CPAP units ($1000 each) of $15,600 were not factored into these since such costs would vary but are given in Table 1. The recurrent per study cost ($86.50) was multiplied by the average number of studies per group, added to the one-time consumables cost, and the total was multiplied by the number of subjects per group. The per patient cost for performing PSMGY at our sleep disorders center is estimated to be $979. This considers cost and depreciation for equipment and space rental, cost of utilities, insurance, administration and personnel, maintenance, and bad debts (nonpaying patients).
|
|

|
|
Table 1. Cost Comparison of Diagnostic Home Studies vs In-Laboratory Polysomnography (PSMGY)*
|
|
|
Mean differences between the 4 subgroups (Table 2 and Figure 2) (OSA-TIT, OSA-NEG, OSA-UNTIT, and UNSAT-DIAG) were compared by analysis of variance for multiple groups. Mean differences between baseline and follow-up parameters including questionnaires and pre–RDI- and post–RDI-machine or pre–RDI- and post–RDI-visual scores were compared by t test for paired data. Throughout the report, the significance level for rejection of the null hypothesis is P<.05. All variance expressed in the text and figures is ±1 SD.
|
|

|
|
Table 2. Baseline Morphometric, Sleep, and Questionnaire Parameters*
|
|
|
RESULTS
The mean age of the 63 subjects (58 men, 5 women) was 51.4 ± 1.4 years; and mean body mass index (calculated as weight in kilograms divided by the square of height in meters), 35.8 ± 1.2 kg/m2. The mean baseline score on the ESS was 15.7 ± 0.6 (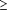 11 is considered symptomatic) and that of the CQ was 39.7 ± 1.0 ( 11 is considered symptomatic) and that of the CQ was 39.7 ± 1.0 (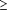 25 is considered symptomatic). Nine subjects (14%) (UNSAT-DIAG) were unable to complete sufficient diagnostic studies to establish or refute a diagnosis of OSA (Figure 2). This was mainly owing to intolerance or removal of the nasal mask, inability to connect the equipment properly after several attempts, or failure to return after an initial unsatisfactory study. Fifty-four subjects (86%) completed adequate diagnostic studies, 45 (83%) of whom met criteria for OSA (RDI-visual score 25 is considered symptomatic). Nine subjects (14%) (UNSAT-DIAG) were unable to complete sufficient diagnostic studies to establish or refute a diagnosis of OSA (Figure 2). This was mainly owing to intolerance or removal of the nasal mask, inability to connect the equipment properly after several attempts, or failure to return after an initial unsatisfactory study. Fifty-four subjects (86%) completed adequate diagnostic studies, 45 (83%) of whom met criteria for OSA (RDI-visual score 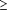 10 respiratory events per hour) and 9 (16%) of whom had an RDI-visual score lower than 10 per hour (OSA-NEG). Of the 45 subjects with OSA, 35 (78%) completed titration studies (OSA-TIT), but 10 were unable to do so (OSA-UNTIT). 10 respiratory events per hour) and 9 (16%) of whom had an RDI-visual score lower than 10 per hour (OSA-NEG). Of the 45 subjects with OSA, 35 (78%) completed titration studies (OSA-TIT), but 10 were unable to do so (OSA-UNTIT).
Of the 35 OSA-TIT subjects, 30 (67% of the 45 subjects diagnosed with OSA) used the machine for 3 weeks or longer, allowing completion of follow-up parameters (ESS, CQ, and multiple sleep latency test), and 5 either dropped out of the study or moved from the area before preliminary treatment follow-up could be done. With few exceptions, morphometric parameters, ESS and CQ scores, and the number of diagnostic studies needed were comparable between groups (Table 1). On average, 1.4 diagnostic studies and 2.4 auto-CPAP titration studies per patient were required to make a diagnosis and reach satisfactory treatment pressures. The correlation coefficient between RDI-machine and RDI-visual scores was 0.85 (P<.002). The RDI-visual scores were systematically higher than RDI-machine scores (Table 3) because visual scoring allowed lowering of the denominator (time) during mask or mouth leaks or off time were subtracted from the total recording time.
|
|

|
|
Table 3. Baseline and Follow-up Parameters From OSA-TIT Group*
|
|
|
Of the 30 subjects who completed auto-CPAP titration at home and kept the machine long enough to evaluate a response to therapy (>3 weeks), the RDI-machine scores fell from 21.2 to 6 respiratory events per hour with treatment (Table 3) while the RDI-visual scores fell from 34.1 to 8.6 respiratory events per hour. The ESS score improved from 16.8 to 10.5 (P<.001), and the CQ score improved from 39.5 to 24.3 (P<.001). Twenty-six OSA-TIT subjects completed 4 or more daytime nap studies at baseline and follow-up. Mean sleep onset latency was 3.7 minutes before treatment and 5.7 minutes after treatment (P<.002). Analysis of this subgroup shows that the baseline and follow-up RDI-visual scores were 32.2 ± 21.0 and 9.1 ± 4.8 respiratory events per hour, respectively. Mean compliance for the group was 4.0 ± 2.1 hours per night. The correlation coefficient for linear regression between hourly compliance and change in baseline to follow-up multiple sleep latency test was 0.12 (P>.99).
Of the 9 UNSAT-DIAG subjects, 2 refused and 7 completed PSMGY. In the latter group, 5 were confirmed with at least 10 or more respiratory events per hour (RDI scores of 51, 11, 98, 60, and 31 per hour, respectively), and 2 were not confirmed for at least 10 respiratory events per hour but were suspected by us of having upper airway resistance syndrome (RDI scores <10 per hour with loud snoring and symptoms of daytime sleepiness).21 Of the 10 OSA-UNTIT subjects, 8 refused to take an auto-CPAP machine home following formal PSMGY and CPAP titration, and 2 dropped out and sought therapy outside of the study. Four of the OSA-TIT subjects were noncompliant with therapy in the first few weeks of home treatment and were asked to return the machines. One subject moved from the study area, seeking treatment elsewhere.
COMMENT
THE NEED FOR ALTERNATIVES TO PSMGY
Based on recent epidemiologic reports, estimates, and ongoing longitudinal studies of various subpopulations (older people, truck drivers, patients at primary care practices), it seems that the prevalence of OSA is much higher than previously suspected.8-9 The standard for the diagnosis and treatment of OSA has been one or more nights of study using in-laboratory PSMGY followed by technician-monitored titration with a nasal CPAP machine, again using PSMGY. As more and more health care practitioners become aware of the symptoms associated with sleep disorders, referrals to sleep laboratories for formal PSMGY should increase. However, PSMGY is labor intensive, may be expensive, may be backlogged owing to sleep laboratory slot availability, may not be readily available to rural patients, and usually entails referral to a sleep specialist. Numerous home monitoring devices have been tested to supplement PSMGY, but sensitivity and specificity vary depending on the device and the examiner.12-14 Home studies have often been espoused as "screening exams" to allow a better selection of patients to be referred for formal PSMGY. However, since an unattended study with normal findings does not preclude apnea, home monitoring cannot be economically satisfactory as a screening examination if it results in the patient undergoing 2 studies instead of 1. Home monitoring needs to serve as a diagnostic device, which when the findings are abnormal can lead to treatment, but when the findings seem normal may require the patient's undergoing PSMGY. In this way it might decrease the expense of diagnosing and treating patients with OSA and perhaps shorten the waiting time from clinical suspicion to treatment.
DIAGNOSIS AND TREATMENT WITH THE SAME DEVICE
Several auto-CPAP machines have been approved for use in the United States and Europe. Within a few years, in-laboratory CPAP titration may be extensively supplemented by home auto-CPAP titration.22-23 Some of these machines have dedicated diagnostic computers that measure airflow, position, and oximetry. Thus, theoretically, the same auto-CPAP machine could be used to establish the presence of apnea, followed on subsequent nights by auto-CPAP titration. Employing this paradigm, our study yielded the following important findings: (1) Fifty-four (86%) of 63 subjects enrolled were able to complete satisfactory, self-applied, home monitoring, and 45 (83%) of these had a diagnosis of OSA established without PSMGY. (2) Of 45 subjects in whom a diagnosis of OSA was established, 35 (78%) were able to be titrated at home by auto-CPAP titration to an adequate pressure to manage their apnea and hypopnea and snoring. (3) Thirty (67%) of the 45 subjects with OSA accepted in-home us of the machine for chronic treatment. (4) There could be substantial savings considering that home studies would have cost about one fourth that of PSMGY monitoring.
We did not use PSMGY as an initial diagnostic tool with direct comparison to the unattended monitoring device for 2 reasons. First, our home monitoring device used a pneumotachograph, which is much more accurate for measuring respiration than a nasal thermistor, routinely used in PSMGY.24-26 We were readably able to examine flow limitation since we could visualize breath-by-breath inspiratory flow morphologic characteristics.25-26 Therefore, it would be difficult to say which should be considered the criterion standard in such a comparison—a semiquantitative nasal thermistor in PSMGY or a more accurate pneumotachograph in this unattended monitoring system. Second, to test the paradigm as postulated, we felt that it was important not to be influenced by the results of the PSMGY but instead to apply historical and unattended CPAP monitoring and titration results to arrive at a diagnosis and therapeutic plan and then request PSMGY in the event of failure or doubt. This particular unattended system was chosen because it could function as both a diagnostic and titration tool, minimizing instruction and familiarization time to the patient by using only 1 device.
CRITIQUE
A criticism of this study is that the low nasal airflow generated by a CPAP pressure of only 3 cm H2O was uncomfortable for some subjects, causing them to abandon diagnostic workup within the study paradigm. Subjects failing for this reason would have been in the UNSAT-DIAG group. Two of the 9 subjects refused PSMGY, and true diagnosis for their condition remains unknown. Two had PSMGY results that showed RDIs of 51 and 11 per hour and accepted home CPAP therapy. Five others had PSMGY results that showed either OSA (RDI scores of 98, 60, and 31 per hour) or as in 2 of these 5, were suggestive of upper airway resistance syndrome.21 But following diagnostic PSMGY, they all refused in-laboratory CPAP titration and may have in general been intolerant of nasal masks. Thus, of the 63 subjects, it is possible that 2 were unable to complete adequate diagnostic studies because of the low diagnostic CPAP pressure required to operate the pneumotachograph.
A contrary hypothesis is that, theoretically, a CPAP pressure of 3 cm H2O eliminated apneas in some subjects during the diagnostic night, concealing an abnormal diagnosis. We believe this to be unlikely in that the usual nasal mask pressure at which upward CPAP titration begins is 5 cm H2O, and few patients are adequately treated at this level. Since it was necessary to have some air flowing over the pneumotachograph for the system to work, as well as to provide the patient with fresh airflow through the occluding nasal mask, this situation was unavoidable. False-negative diagnoses missed because of the diagnostic CPAP pressure of 3 cm H2O would have appeared in the OSA-NEG group (n = 9). Of these, 3 subjects refused PSMGY, 2 had mild apnea with in-laboratory RDIs of 17 and 15 per hour, respectively, and 4 had normal findings from PSMGY. Thus, it is possible that up to 5 diagnoses (2 subjects with normal findings and 3 refusing PSMGY) could have been missed because of the 3 cm H2O CPAP pressure concealing apnea. It is more likely that intrasubject variability was the cause of missing the diagnosis in these subjects.11 Nevertheless, by the algorithm, such patients (subjects with normal findings) would have been referred for PSMGY and thus been diagnosed by a thermistor or other airflow detection device in the laboratory.
NASAL CPAP INTOLERANCE
The initial refusal to accept complete CPAP titration or take the machine home is compatible with published in-laboratory CPAP compliance reports.27-29 For example, Waldhorn et al27 report that 19 (15%) of 125 of patients with OSA refused a home nasal CPAP trial or did not tolerate nasal CPAP titration in the laboratory. Krieger et al28 report that 153 (21%) of 728 patients with OSA refused to accept home CPAP therapy. Schweitzer et al29 report that an overall 33 (24%) of 133 patients with OSA refused home CPAP therapy (either titration or taking the machine home). Furthermore, 16 (12%) of 133 patients who tolerated the CPAP titration in the laboratory refused to take the machine home. Thus, our 18% home auto-CPAP refusal rate in patients with proven OSA appears to be compatible with published figures on CPAP intolerance and not directly owing to unattended home diagnostic or treatment monitoring.
The estimated cost of all home diagnostic studies was $22,694 (Table 1), which includes diagnostic PSMGY in the 7 failed home study patients who agreed to undergo PSMGY (Figure 2). Figuring that all of these subjects would require only 1 diagnostic PSMGY in our sleep disorders center at a cost of $979 per patient, that diagnostic method would have cost $61,677 for the same subjects. The estimated cost of all home titration studies was $7093, allowing the same costs as a diagnostic study. Figuring that all of these subjects would require only 1 CPAP titration by PSMGY at the same cost of $979 per patient, it would have cost $39,160 for the same subjects. The total cost for all unattended home studies would have been $29,787 and for PSMGY, $100,837. It is difficult to compare the exact cost of home monitoring with PSMGY since costs will vary with geographic location, employee salaries, physician charges, cost for consumables, etc. We have itemized our costs so that the reader might substitute their own local costs to examine the cost-savings of home monitoring vs PSMGY. We did not factor in the cost of the home monitoring equipment because the purchase cost of sleep laboratory equipment is not factored into PSMGY costs. Adding the cost of a 3-patient monitoring system ($15,600) to the total cost of monitoring the patients in this study would come to less than $40,000.
THE HOME OSA DIAGNOSIS/TREATMENT ALGORITHM
The purpose of this study was not to abrogate the role of PSMGY or the sleep specialist in the workup and treatment of patients with OSA, but to create a more selective PSMGY referral population. Just as patients with nonspecific chest pain, arthritis, or asthma have a recognizable constellation of signs and symptoms, the signs and symptoms of patients with OSA may be typical and classic. Once suspected, the clinician must confirm the diagnosis and initiate treatment by an accurate, cost-effective method. Current practice takes the diagnosis and treatment of OSA out of the hands of the primary physician and transfers it to the sleep laboratory. This may in part discourage the primary care giver from learning more about basic sleep disorders, such as OSA.
This study did not ask what level of physician training is required to interpret these study results (since this study was done by subspecialists engaged in the practice of sleep medicine), nor did it provide data as to the application of this algorithm to the family practice or primary care setting. To determine such would require a prospective study applied to the primary care environment. However, the technical requirement for operating this equipment is well within the grasp of most nurses and office personnel. The skills for taking an accurate sleep history and learning to deal with simple CPAP problems are within reach of the family medicine practitioner, especially with such tools as the ESS and CQ available to quantitate symptoms.
If symptomatic improvement does not occur within a few weeks of initiating CPAP therapy and the patient is shown by the CPAP hourly use meter to be compliant, an algorithm similar to that followed in this study can be instituted. That is, referral for PSMGY should occur if the patient has (1) normal study results (<10 apneas and/or hypopneas per hour), (2) an unsatisfactory diagnostic study (missing data, major mask or mouth leaks precluding scoring, short recording time, etc), (3) is unable to be titrated to an acceptable CPAP pressure, (4) refused to accept the auto-CPAP machine for home use, or (5) remained symptomatic (sleepy) after several weeks of objectively verified home auto-CPAP use. Were this clinical paradigm to be applied to home monitoring and auto-CPAP titration, then patients with complicated disorders, such as OSA plus periodic leg movements (restless legs syndrome), OSA plus narcolepsy, or patients demanding more individual attention in adjusting to the equipment, would undergo in-laboratory PSMGY and treatment. In a recent report,30 a rural primary care facility without a sleep specialist increased the rate of diagnosis of sleep disorders in its patient population of 14,330 from 0.27% to 2.1% (294 cases) over a 1-year period by increased alertness and interest of its physicians in this new area of medicine.
Current American31 and Australian32 consensus panels recommend that every patient with suspected sleep apnea syndrome receive formal in-laboratory PSMGY. British guidelines have not followed this and state that oximetry alone or oximetry plus video recording is sufficient to diagnose many patients.33 The latter position is supported in a recent report comparing oximetry with PSMGY.34 We believe that in the future, in-home unattended diagnosis and CPAP titration will be common, especially in more severe patients with classic symptoms and in patients with limited access to PSMGY. Much of the initial testing could be done by primary care physicians, reserving the more difficult diagnosis and treatment cases for sleep specialists.
AUTHOR INFORMATION
Accepted for publication June 28, 1999.
Reprints: Eugene C. Fletcher, MD, Division of Respiratory, Critical Care, and Environmental Medicine, University of Louisville School of Medicine, Ambulatory Care Building, Room A3L01, 530 S Jackson St, Louisville, KY 40202.
From the Department of Medicine and the Division of Respiratory, Critical Care, and Environmental Medicine, Louisville Veterans Affairs Medical Center, University of Louisville School of Medicine, Louisville, Ky.
REFERENCES
 |  |
1. Fletcher EC. Sleep apnea syndrome. In: Kelley WN, ed. Kelley's Textbook of Internal Medicine. 3rd ed. Philadelphia, Pa: JB Lippincott Co; 1997:2078-2085.
2. Fletcher EC. Primary hypertension and obstructive sleep apnea: fact and theory. Am J Med. 1995;98:118-128.
FULL TEXT
|
ISI
| PUBMED
3. Partinen M, Palomaki H. Snoring and cerebral infarction. Lancet. 1985;2:1312-1326.
4. Franklin KA, Milsson JB, Sahlin C, Naslund E. Sleep apnea and nocturnal angina. Lancet. 1995;345:1085-1087.
FULL TEXT
|
ISI
| PUBMED
5. Shepard JW Jr. Hypertension, cardiac arrhythmias, myocardial infarction, and stroke in relation to obstructive sleep apnea. Clin Chest Med. 1992;13:437-458.
ISI
| PUBMED
6. He J, Kryger M, Zorick F. Mortality and apnea index in obstructive sleep apnea. Chest. 1988;89:331-334.
7. Partinen M, Guilleminault C. Mortality of patients with obstructive sleep apnea syndrome: a follow-up study. Chest. 1988;94:1200-1204.
FREE FULL TEXT
8. Young T, Palta M, Dempsey J. The occurrence of sleep-disordered breathing among middle-aged adults. N Engl J Med. 1993;328:1230-1235.
FREE FULL TEXT
9. Smith PL, Hudgel DW, Olson LG. Indications and standards for use of nasal continuous positive airway pressure (CPAP) in sleep apnea syndromes. Am J Respir Crit Care Med. 1994;150:1738-1745.
ISI
| PUBMED
10. Sullivan CE, Issa FG, Berthon-Jones M, Eves L. Reversal of obstructive sleep apnoea by continuous positive airway pressure applied through the nares. Lancet. 1981;1:862-865.
ISI
| PUBMED
11. Mosko SS, Dickel MJ, Ashurst J. Night-to-night variability in sleep apnea and sleep-related periodic leg movements in the elderly. Sleep. 1988;11:340-348.
ISI
| PUBMED
12. White DP, Gibb TJ, Wall JM, Westbrook PR. Assessment of accuracy and analysis time of a novel device to monitor sleep and breathing in the home. Sleep. 1995;18:115-126.
ISI
| PUBMED
13. Ferber R, Millman R, Coppola M. ASDA Standards of Practice: portable recording in the assessment of obstructive sleep apnea. Sleep. 1994;17:378-392.
ISI
| PUBMED
14. ASDA Standards of Practice: practice parameters for the use of portable recording in the assessment of obstructive sleep apnea. Sleep 1994;17:372-377.
ISI
| PUBMED
15. Teschler H, Berthon-Jones M, Thompson AB. Automated continuous positive airway pressure applied titration for obstructive sleep apnoea. Am J Respir Crit Care Med. 1996;154:734-740.
ABSTRACT
16. Miles LE, Buschek GD, McClintock DP. Development and application of automatic nasal CPAP calibration procedures for use in the unsupervised home environment. Sleep. 1993;16(suppl):S118-S119.
17. Scharf MB, Brannen DE, McDannold MD, Berkowitz DV. Computerized adjustable versus fixed NCPAP treatment of obstructive sleep apnea. Sleep. 1996;19:491-496.
ISI
| PUBMED
18. Johns MW. A new method for measuring daytime sleepiness: the Epworth Sleepiness Scale. Sleep. 1991;14:540-545.
ISI
| PUBMED
19. Kump K, Whalen C, Tishler PF. Assessment of the validity and utility of a sleep-symptom questionnaire. Am J Respir Crit Care Med. 1994;150:735-741.
ABSTRACT
20. Stradling JR, Barbour C, Pitson DJ, Davies RJO. Automatic nasal continuous positive airway pressure titration in the laboratory: patient outcomes. Thorax. 1997;52:72-75.
FREE FULL TEXT
21. Guilleminault C, Stoohs R, Kim Y, Chervin R, Black J, Clerk A. Upper airway sleep-disordered breathing in women. Ann Intern Med. 1995;122:493-501.
FREE FULL TEXT
22. Berthon-Jones M. Feasibility of a self-setting CPAP machine. Sleep. 1993;16(suppl):S120-S123.
23. Sharma S, Wali S, Pouliot Z. Treatment of obstructive sleep apnea with a self-titrating continuous positive airway pressure (CPAP) system. Sleep. 1996;19:497-501.
ISI
| PUBMED
24. Farre R, Montserrat JM, Rotger M, Ballester E, Navajas D. Accuracy of thermistors and thermocouples as flow-measuring devices for detecting hypopnoeas. Eur Respir J. 1998;11:179-182.
FREE FULL TEXT
25. Condos R, Norman RG, Krishnasamy I, Peduzzi N, Goldring RM, Rapoport D. Flow limitation as a noninvasive assessment of residual upper-airway resistance during continuous positive airway pressure therapy of obstructive sleep apnea. Am J Respir Crit Care Med. 1994;150:475-80.
ABSTRACT
26. Montserrat JM, Ballester E, Olivi H, et al. Time-course of stepwise CPAP titration. Am J Respir Crit Care Med. 1995;152:1854-1859.
ABSTRACT
27. Waldhorn RE, Herrick TW, Nguyen MC. Long-term compliance with continuous positive airway pressure therapy of obstructive sleep apnea. Chest. 1990;97:33-38.
FREE FULL TEXT
28. Krieger J, Kurtz D, Petiau C. Long-term compliance with CPAP therapy in obstructive sleep apnea patients and in snorers. Sleep. 1996;19(suppl):S136-S143.
29. Schweitzer PK, Chambers GW, Birkermeier N. Nasal continuous positive airway pressure (CPAP) compliance at six, twelve and eighteen months [abstract]. Sleep Res. 1987;16:186.
30. Ball EM, Simon RD Jr, Tall AA, Banks MB, Nino-Murcia G, Dement WC. Diagnosis and treatment of sleep apnea within the community. Arch Intern Med. 1997;157:419-424.
FREE FULL TEXT
31. Phillipson EA, Remmers JE. Indications and standards for cardiopulmonary sleep studies. Am Rev Respir Dis. 1989;139:559-568.
ISI
| PUBMED
32. McAvoy RD. Guidelines for Respiratory Sleep Studies. Sydney, Australia: Thoracic Society of Australia and New Zealand; 1988.
33. British Thoracic Society. Facilities for the diagnosis and treatment of abnormal breathing during sleep. BTS News. 1990;5:7-10.
34. Douglas NJ, Thomas S, Jan MA. Clinical value of polysomnography. Lancet. 1992;339:347-350.
FULL TEXT
|
ISI
| PUBMED
RELATED ARTICLE
The Archives of Family Medicine Continuing Medical Education Program
Arch Fam Med. 2000;9(2):175-177.
FULL TEXT
THIS ARTICLE HAS BEEN CITED BY OTHER ARTICLES
Treatment of obstructive sleep-disordered breathing with positive airway pressure systems
Pevernagie et al.
ERR 2007;16:125-131.
ABSTRACT
| FULL TEXT
Alternative Methods of Titrating Continuous Positive Airway Pressure: A Large Multicenter Study
Masa et al.
Am. J. Respir. Crit. Care Med. 2004;170:1218-1224.
ABSTRACT
| FULL TEXT
|