Nascent Transcripts
Emerging concepts from the recent literature
Fluoxetine Causes Depression (of T-type Calcium Channels)
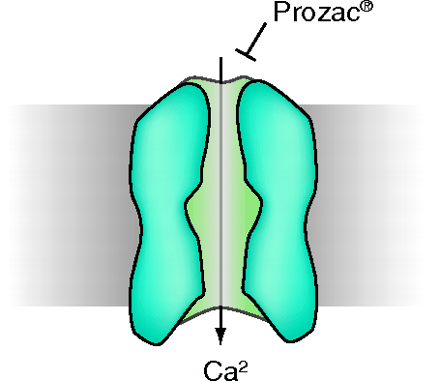
Fluoxetine (Prozac®) is a psychoactive drug widely prescribed in depression. The therapeutic action of fluoxetine results primarily from the inhibition of serotonin reuptake. Recent research shows that fluoxetine also inhibits a variety of voltage-activated K+, Na+, and Ca2+ channels. Traboulsie et al. now report that low voltage–activated T-type Ca2+ channels (T-channels) are inhibited by micromolar concentrations of fluoxetine and its active metabolite norfluoxetine. T-channels play important roles in neuronal firing (e.g., in sleep and epilepsy), and these new data suggest that fluoxetine-inhibition of T-channels may significantly impair neuron excitability. Significantly, fluoxetine preferentially blocks T-channels in the inactivated state, which corresponds to their status in physiological conditions. Inhibition of T-channels thus appears as a novel mechanism by which fluoxetine is pharmacologically active and may account for some of the clinical effects in treated patients as well as side effects. Fluoxetine-inhibition of T-channels should therefore be taken into account in further studies of the pharmacological properties of this widely prescribed antidepressant. [
.] —J Bockaert, IGF, CNRS, France
Gemfibrozil Glucuronide Is a Mechanism-Based Inactivator of CYP2C8
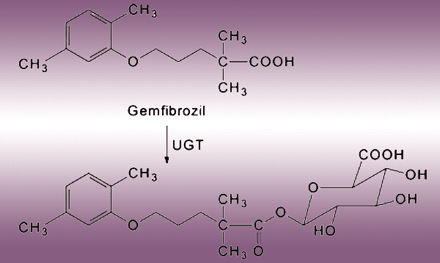
Prediction of drug–drug interactions from in vitro data is an important goal of drug development, yet sometimes these predictions fall short on accuracy. In the January issue of Drug Metabolism and Disposition, Ogilvie et al. explore why, in vitro, gemfibrozil inhibits CYP2C9 to a greater extent than it does CYP2C8, and yet the opposite is observed in vivo. Using human liver microsomal preps and both gemfibrozil and gemfibrozil glucuronide as substrates, these investigators determine the ability of both compounds to inhibit CYP2C9 and CYP2C8. Interestingly, gemfibrozil glucuronide is metabolized by CYP2C8 to a reactive intermediate that inactivates CYP2C8, but not CYP2C9, in a mechanism-based, irreversible fashion. Subsequent oxidative metabolism of a glucuronide conjugate (e.g., diclofenac) has been described previously, but this is the first instance of the formation of a mechanism-based inactivator from a glucuronide. These results explain the gemfibrozil inhibition paradox and stress that additional cautions are required when predicting in vivo drug–drug interactions from in vitro data. [
.] —TS Tracy, University of Minnesota
At Long Last, an Mrp2-Null Mouse
Multidrug resistance–associated protein 2 (Mrp2), a member of the ATP-binding cassette (ABC) transporter family, is an organic-anion export pump expressed in several barrier and excretory tissues. In humans, MRP2 mutations cause the autosomal recessive Dubin-Johnson syndrome, resulting in mild conjugated hyperbilirubinemia. To date, Mrp2 function in vivo has been studied in two rat models. In the May issue of JPET, however, Chu et al. report the production and characterization of Mrp2−/− mice, which, similar to Mrp2−/− rats, exhibit greatly reduced hepatic excretion of bilirubin glucuronide and glutathione. Whereas Mrp2−/− rats compensate in part for loss of Mrp2 by inducing the expression of hepatic Mrp3, Mrp2−/− mice appear to increase the expression of Mrp4 in liver and kidney. These mice will become a valuable tool in the study of Mrp2 in drug absorption and distribution and compensatory mechanisms. [
.] —DS Miller, NIH/NIEHS
Energy-Sparing Inhibitors of Metalloproteinases

Matrix metalloproteinases (MMPs) are thought to mediate angiotensin II (AII)-induced diastolic stiffening, which is a characteristic of cardiac failure. Paolocci et al. have now investigated whether the amelioration of stiffening, through MMP inhibition, arises primarily from matrix remodeling or from the sparing of high-energy phosphate species (e.g., ATP and phosphocreatine). Dogs were administered AII and subjected to tachypacing (i.e., increased beating of the heart) , which activated MMP9 and MMP2, exacerbated diastolic stiffening, and reduced the concentrations of ATP and phosphocreatine while increasing free ADP, AMP catabolites, and lactate. In dogs pretreated with an MMP inhibitor (PD166793), the AII-induced stiffening was prevented and most of the losses in high energy phosphate species were attenuated, in part, by inhibiting the activity of AMP-deaminase. Thus, MMP activation may influence cardiac diastolic function (via high energy phosphate metabolism) by mechanisms beyond the “simple” regulation of extracellular matrix. Moreover, the findings highlight a potential major role for increased AMP deaminase activity in diastolic dysfunction. [
.] —R Levi, Weill Medical College
- © American Society for Pharmacology and Experimental Theraputics 2006



